|
Case Report
Sonidegib: Real-life experience with Hedgehog inhibitor for the treatment of locally advanced basal cell carcinoma
1 Unità Operativa Complessa Oncologia, Ospedale “S. Vincenzo”, Taormina (ME), Sicily, Italy
Address correspondence to:
Alessia Dottore
Alessia Dottore, Unità Operativa Complessa Oncologia, Ospedale “S. Vincenzo”, Taormina (ME), Sicily,
Italy
Message to Corresponding Author
Article ID: 100134Z10AD2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Dottore A, Briguglio R, Priolo D, Ferraù F. Sonidegib: Real-life experience with Hedgehog inhibitor for the treatment of locally advanced basal cell carcinoma. J Case Rep Images Oncology 2024;10(2):6–9.ABSTRACT
Introduction: Sonidegib, a Hedgehog inhibitor approved for the treatment of advanced basal cell carcinoma (BCC), reported an objective response rate of 60.6% (central review) and 74.2% (investigator review) and a median duration of response of 26.1 months in the pivotal trial BOLT. The therapy showed an excellent tolerability profile. The most frequent adverse events are muscle spasms, dysgeusia, alopecia, nausea, increased creatine kinase, weight loss and fatigue.
Case Report: This article reports the case of a neglected patient in which sonidegib rapidly led to an important tumor mass reduction and improvement of quality of life without of adverse effects.
Conclusion: The peculiar efficacy and tolerability of sonidegib, in addition to representing a current gold standard for the treatment of the advanced disease, allow us to hypothesize its use on a larger scale in a modern multidisciplinary strategy (delete with surgery and radiotherapy).
Keywords: Basal cell carcinoma, Real-life experience, Sonidegib
Introduction
Sonidegib, a Hedgehog Inhibitor (HHI) approved for the treatment of advanced basal cell carcinoma (BCC), reported an objective response rate of 60.6% (central review) and 74.2% (investigator review) and a median duration of response of 26.1 months in the pivotal trial BOLT [1]. The therapy showed an excellent tolerability profile. The most frequent adverse events are muscle spasms, dysgeusia, alopecia, nausea, increased creatine kinase, weight loss and fatigue.
Case Report
We present a case report of a 69-year-old woman with a social isolation and rejection of medical treatment, with past medical history of anxiety and depression.
Her story began with a recurrent lesion in the upper right eyelid that was misdiagnosed and surgically treated as a chalazion. Photographic documentation, provided by her caregiver in June 2020, showed a large lesion affecting the right eyelid and the base of the nose. At that time, the patient refused any diagnostic and therapeutic intervention.
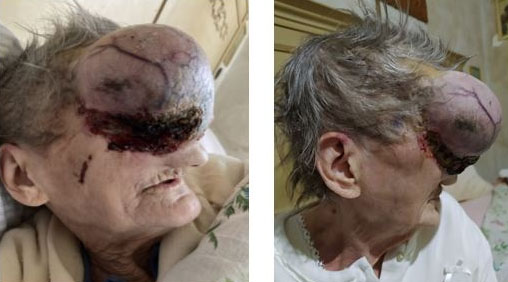
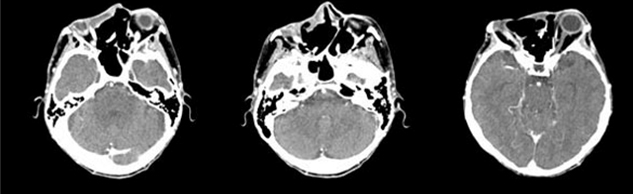
In December 2020, the patient showed a very extensive lesion (delete dome-shaped) measuring 12 × 5 cm in diameter that affected the right orbit, majority of the forehead and nose, and reaching to the left orbit. The mass presented ulceration and neovascularization. Due to pain and general malaise, the patient finally accepted to undergo head and maxillofacial computed tomography (CT) scan that showed wide destruction of the surrounding tissues, including bone structures of nose, right jaw, right orbit, anterior cranial fossa, pterygopalatine fossa, and right infratemporal fossa (Figure 1).
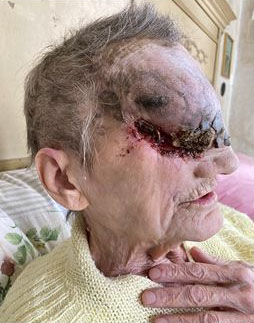
In March 2021, the patient reported for the first time to our center with intense pain and swelling in the right half of the face, diffuse rachialgia, and difficult walking. The mass showed further enlargement with a bleeding ulcer at the base (Figure 2); therefore, the patient allowed a biopsy that confirmed an infiltrating scleroderma-like BCC with baso-squamous features. Since the size and location of the lesion and the concrete risk of hemorrhagic and neurological consequences made any loco-regional approach unfeasible (surgery or radiotherapy), Hedgehog inhibitor therapy was recommended. Sonidegib 200 mg orally once daily was prescribed.
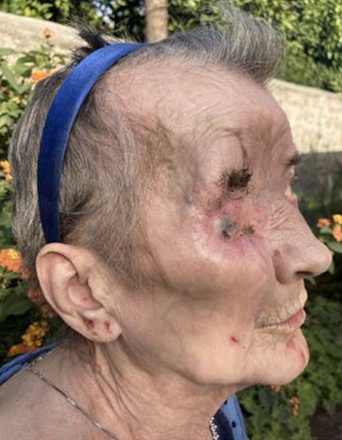
Sonidegib was well tolerated and led to reduction of pain, burning and itching from the first days of treatment. After only 37 days of therapy, the lesion was significantly reduced in size and flattened (Figure 3). In subsequent weeks, there was a constant progressive reduction of the tumor mass. After four months of therapy the lesion was flat (Figure 4) and, as shown on head and maxillofacial computed tomography (CT) scan (Figure 5), the process of destruction of surrounding tissues stopped almost completely.
Discussion
The management of the patient with BCC that cannot be controlled with surgery and/or radiotherapy has seen in recent years a profound shift allowed by the significant results of medical therapy with Hedgehog inhibitors. Sonidegib combined the brilliant cytoreductive activity and an optimal tolerability (as reported in BOLT trial [2]). Our case report show a time to response lower than time to response in the BOLT trial. This “speeds” of response to treatment.
This “speeds” of response to treatment can be found in many case reports reported in the literature [3],[4],[5]. The good tolerability is of particular importance for this subset of patients: they are often elderly and with important comorbidities. In case of adverse events, it’s possible the switch to alternating daily administration (200 mg every other day) before to consider stop treatment. Moreover, sonidegib is for oral use: the patient takes the drug at home without the need to go to hospital. In our case, it’s relevant to highlight that sonidegib had the ability to rescue a neglected patient who had previously refused any other type of therapeutic intervention, allowing her to resume social life and significantly improving quality of life. Finally, it is worthy of note that sonidegib was effective on a BCC with baso-squamous features, as recently reported also by Toffoli et al. [6].
Conclusion
In conclusion, the peculiar efficacy and tolerability of sonidegib, in addition to representing a current gold standard for the treatment of the advanced disease, allow us to hypothesize its use on a larger scale in a modern multidisciplinary strategy.
REFERENCES
1.
Gutzmer R, Robert C, Loquai C, et al. Assessment of various efficacy outcomes using ERIVANCE-like criteria in patients with locally advanced basal cell carcinoma receiving sonidegib: Results from a preplanned sensitivity analysis. BMC Cancer 2021;21(1):1244. [CrossRef]
[Pubmed]

2.
Dummer R, Guminski A, Gutzmer R, et al. The 12-month analysis from Basal Cell Carcinoma Outcomes with LDE225 Treatment (BOLT): A phase II, randomized, double-blind study of sonidegib in patients with advanced basal cell carcinoma. J Am Acad Dermatol 2016;75(1):113–25.e5. [CrossRef]
[Pubmed]

3.
Trabelsi S, Khidher F. Rapid onset of response to sonidegib for multiple facial basal cell carcinomas during COVID-19 pandemic. Dermatol Ther 2022;35(4):e15317. [CrossRef]
[Pubmed]

4.
Tarantino V, Zavattaro E, Veronese F, Gironi LC, Savoia P. Rapid and exceptional response to sonidegib in a patient with multiple locally advanced basal cell carcinomas. Anticancer Drugs 2021;32(4):465–68. [CrossRef]
[Pubmed]

5.
Dummer R, Guminksi A, Gutzmer R, et al. Long-term efficacy and safety of sonidegib in patients with advanced basal cell carcinoma: 42-month analysis of the phase II randomized, double-blind BOLT study. Br J Dermatol 2020;182(6):1369–78. [CrossRef]
[Pubmed]

6.
Toffoli L, Agozzino M, di Meo N, Zalaudek I, Conforti C. Locally advanced basosquamous carcinoma: Our experience with sonidegib. Dermatol Ther 2022;35(6):e15436. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Alessia Dottore - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Roberta Briguglio - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Domenico Priolo - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Francesco Ferraù - Conception of the work, Design of the work, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Alessia Dottore et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.