|
Case Series
Safe and effective use of cabozantinib and nivolumab in stabilizing disease progression of tissue sarcoma after multiple lines of standard therapy
1 Resident Physician, Department of Internal Medicine, Brooke Army Medical Center, San Antonio, Texas, USA
2 Staff Pathologist, Department of Pathology, Brooke Army Medical Center, San Antonio, Texas, USA
3 Fellow Physician, Department of Hematology/Oncology, Brooke Army Medical Center, San Antonio, Texas, USA
4 Staff Hematologist/Oncologist, Department of Hematology/Oncology, Brooke Army Medical Center, San Antonio, Texas, USA
Address correspondence to:
My Linh D Vu
MD, 3551 Roger Brooke Dr, San Antonio, TX 78219,
USA
Message to Corresponding Author
Article ID: 100123Z10KS2023
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Vu MLD, Berman A, Kao E, Stoll K, Lee L. Safe and effective use of cabozantinib and nivolumab in stabilizing disease progression of tissue sarcoma after multiple lines of standard therapy. J Case Rep Images Oncology 2023;9(2):1–6.ABSTRACT
Introduction: Soft-tissue sarcomas (STS) are a rare and varied group of tumors with limited treatment options. In recent years, immune checkpoint inhibitors have demonstrated efficacy in an increasing number of solid tumors. Nivolumab, a PD1-inhibitor, demonstrates improved overall survival (OS) and progression-free survival (PFS) in renal cell carcinoma, melanoma, and gastrointestinal stromal tumor (GIST). The tyrosine kinase inhibitor (TKI), cabozantinib, demonstrates an antitumor effect in osteosarcoma and Ewing sarcoma.
Case Series: Here we describe two patients with recurrent and refractory STS. Both patients failed multiple lines of conventional therapy including neoadjuvant radiation, surgical resection, and palliative chemotherapy before achieving partial response with combination cabozantinib and nivolumab.
Conclusion: These cases demonstrate the safety and efficacy of using combination nivolumab and cabozantinib in treatment of STS warranting further investigation of immunotherapy treatment.
Keywords: Cabozantinib, Immunotherapy, Metastatic sarcoma, Nivolumab
Introduction
Soft-tissue sarcomas (STS) are a rare and heterogeneous group of mesenchymal malignant tumors accounting for <1% of adult cancers representing more than 100 histopathologic subtypes and 60 distinct diagnoses, making treatment challenging [1]. Despite aggressive treatment, the five-year survival rates for STS vary between 15–81% [2] . Standard of care consists of neoadjuvant and adjuvant cytotoxic chemotherapy with anthracycline-based regimens, such as doxorubicin, ifosfamide, and mesna (AIM). While anthracycline-based regimens are recommended for initial therapy, second-line treatment is targeted to specific sarcoma types given the wide array of mutational variations and subtypes [3].
Undifferentiated pleomorphic sarcoma (UPS) represents approximately 20% of the STS subtypes [4]. It is an aggressive pleomorphic malignancy without identifiable differentiation or specific morphologic immunohistochemistry markers for cell lineage. As such, it is a diagnosis of exclusion. Undifferentiated pleomorphic sarcoma tumors most commonly occur in the head, neck, retroperitoneum, and extremities [5]. In the metastatic setting, despite cytotoxic agents, survival rates for UPS continue to hover between 12 and 18 months. Similarly, spindle cell sarcoma (SCS) is a similar subtype of STS characterized by spindle-shaped morphology on pathology. Spindle cell sarcoma tends to occur more in patients >40 years old with an almost 2-fold incidence in males but treatment methods are similar to UPS with neoadjuvant chemotherapy and radiation followed by surgical removal [6]. Previously, prognostic factors for both relied heavily on tumor grade location and size at diagnosis [7]. More recently, molecular alterations identified by Next Generation Sequencing found strong correlations with gene mutations in signaling pathways involving the cell cycle, PI3K/mTOR, and RAS/MAPK signaling pathway in resected UPS [8]. Another study evaluating the genomic profile of over 7000 STS cases found CDKN2A deletion was reflective of poor prognosis with significantly worse overall survival with hazard ratio of 2.7 [9].
With the development of immuno-oncology and a further understanding of genomics, immunotherapy has shown promising results in various solid tumors. The Pazopanib explored in soft-tissue sarcoma—a phase 3 study, also known as the PALETTE trial, showed promising results with targeted immunotherapy such as pazopanib, a tyrosine kinase inhibitor, in the subsequent management of STS [10]. CABONE, a phase 2 trial, suggests Cabozantinib can achieve up to six months of non-progression in Ewing sarcoma [11]. Checkpoint inhibitors such as nivolumab and pembrolizumab, both anti-programmed cell death one monoclonal antibody (anti-PD1), are FDA-approved for several solid tumors including melanoma, non-small cell lung cancer, prostate cancer, and renal cell carcinoma, among others, with encouraging results [12],[13]. A phase 3 trial for metastatic renal cell carcinoma, CheckMate 9ER, showed a median progression-free survival of 16.6 months compared to 8.3 months, and an overall survival rate at 12 months of 85.7% compared to 75.6% in patients treated with combination nivolumab and cabozantinib versus sunitinib monotherapy, respectively [14]. Furthermore, the combination of nivolumab and cabozantinib showed only 5.6% of patients discontinued both drugs due to adverse events, suggesting a promising safety profile [14]. A post-hoc review of CheckMate 9ER suggest most adverse events were manageable symptomatically or with adjustment in dosages since half-life of nivolumab and cabozantinib is 25 days and 99 hours, respectively [15].
Though STS continues to be difficult to treat, Movva et al. assessed PD-L1 expression in UPS cases and demonstrated that 70% had high PD-L1 expression [1]. Additionally, Wunder et al. recently demonstrated better overall survival with hazard ratio of 6.27 in patient exhibiting high compared to low PD-L1 level [16]. The strong immune presence offers the promise of a target for immune therapy and justifies their use, particularly for salvage therapy for progressive, metastatic disease. Here, we discuss the use of nivolumab and cabozantinib in two cases of refractory, progressive STS by demonstrating both safety and effective disease response.
Case Series
Case 1
A 39-year-old Caucasian woman presented with a progressively enlarging painful mass on the left knee. Computed tomography (CT) imaging demonstrated a 6.2 × 3.5 × 5.3 cm bony lesion in the lateral femoral condyle. Pathology identified a malignant hypercellular spindle-cell lesion with a brisk mitotic rate favoring high-grade fibrosarcoma, osteosarcoma, or differentiated component chondrosarcoma. Genomic testing revealed a loss of CDKN2A/B and a mutation of PTPN11, which is found in the RAS/MAPK signaling pathway. Initial histologic staining was significant for TLE-1. SYT gene re-arrangement was negative by fluorescence in situ hybridization (FISH) and molecular testing was BCOR negative, therefore not consistent with synovial sarcoma or Ewing-like sarcoma, respectively. Positron emission tomography-computed tomography (PET-CT) did not detect distant disease, and she was diagnosed with stage IIA, pT1a N0 M0 G3 high-grade spindle cell sarcoma (Figure 1). She underwent neoadjuvant methotrexate, doxorubicin, and cisplatin followed by resection. Surgical tissue showed only 20% necrosis suggesting a high risk of recurrence therefore she received 4 cycles of adjuvant ifosfamide (10 gm/m2).
A year following her initial diagnosis, she presented with chest wall pain. A CT chest revealed a right lower lobe pleural-based mass measuring up to 2.7 × 1.1 × 2.9 cm suggesting recurrent disease. A biopsy of the lesion confirmed recurrent disease and she underwent surgical resection with curative intent. However, at the time of surgery, diaphragmatic involvement was discovered, preventing full removal. Tissue confirmed a similar histologic appearance with an evolved de-differentiation raising the possibility of UPS. She underwent 9 cycles of regorafenib before discontinuing for progressive disease with enlarging solid pleural and chest wall masses. Therapy with gemcitabine and docetaxel was initiated but complicated by severe toxicities including myelosuppression requiring hospitalization. An attempt with monotherapy with Gemcitabine continued to have significant toxicities and progressive disease with a greater than 20% increase in tumor burden.
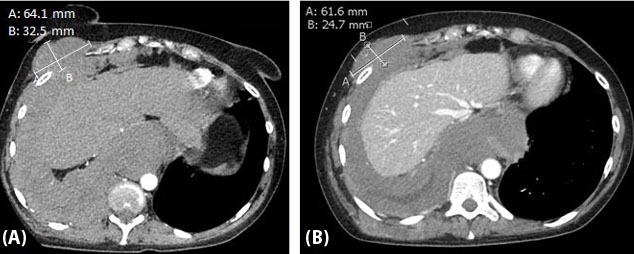
She was referred to and evaluated for clinical trials at a cancer center of excellence, for which a phase 1 oral checkpoint inhibitor was offered even though her PD-L1 tumor proportion score of 0%. She ultimately opted to continue therapy locally and was started on similar treatment with nivolumab and cabozantinib. While on combined therapy, she received surveillance imaging at three-month intervals. She tolerated combined nivolumab and cabozantinib therapy well with only grade 1 toxicity of diarrheal symptoms that was manageable with supportive therapy. Remarkably, first interval imaging demonstrated a decrease in the size of a right mediastinal lymph, dominant right chest wall nodule, and dominant right lung nodule decreased from 6.4 × 3.3 cm to 6.2 × 2.5 cm, representing an approximate 25% size reduction. She achieved a maximal response of approximately 30% decrease in tumor burden and stable disease achieved for almost eight months until the disease again progressed (Figure 2). She then opted to discontinue treatment and pursue hospice and passed shortly after.
Case 2
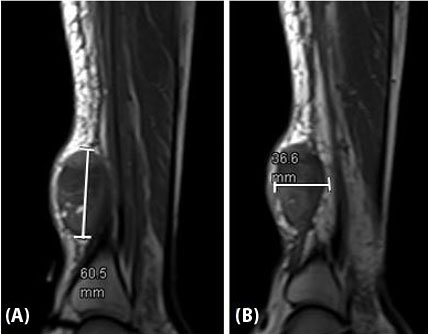
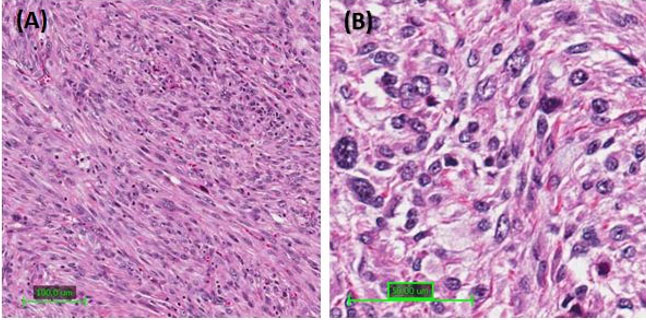
A 53-year-old African American male presented with an enlarging mass on the right lower leg. Initial X-rays noted a 0.9 cm soft tissue mass that appeared suspicious for intramuscular hematoma on ultrasound. Histology of surgically drained tissue was concerning for malignancy. Magnetic resonance imaging (MRI) showed a 6.1 × 4.8 × 2.6 cm mass anterior to the distal tibia ultimately staged as cT2 N0 M0 G3, resulting in Stage IIIA diagnosis (Figure 3). Immunohistochemistry staining demonstrated UPS given no clear line of differentiation and high Ki67 expression associated with rapid cell proliferation and growth (Figure 4). Genomic testing revealed a FOXP1-TL1X fusion along with mutation of TP53, HRAS, and loss of NF1.
He received neoadjuvant radiation and resection with negative margins. No adjuvant therapy was administered, and surgical tissue showed 5% necrosis. Surveillance PET-CT seven months later detected a recurrence of disease with new pulmonary lesions. He underwent palliative AIM for 6 cycles but discontinued when disease progressed on imaging. Pathology confirmed recurrent disease with similar histologic characteristics and PDL1 tumor portion score was 30%. He received palliative Docetaxel/Gemcitabine for 6 cycles until disease again progressed with an over 30% size increase of the pulmonary nodules in both upper lobes and right middle lobe. He quickly then progressed through pazopanib and trabectedin in the span of three months and his disease burden increased over 40%.
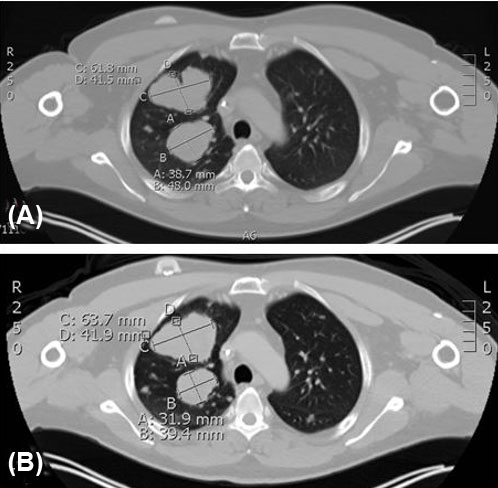
Considering his PD-L1 tumor portion score of 30%, nivolumab was initiated. Shortly after, interval CT showed possible progression in size of pulmonary nodules, prompting the addition of cabozantinib. The patient had minimal side effects on combination nivolumab and cabozantinib only to include palmar plantar erythrodysesthesia that was well managed with topical emollients and steroids. Positron emission tomography-computed tomography imaging showed partial/mix response of dominant pulmonary nodules, but mediastinal lymph nodes reduced by greater than 30% (Figure 5). He achieved disease stability on combination nivolumab/cabozantinib therapy with symptomatic improvement of cough and shortness of breath up to nine months before disease again progressed. He was trialed briefly on one cycle of regorafenib with poor tolerance and no benefit, therefore he transitioned electively to hospice care.
Discussion
In both cases, stable disease and even partial response was achieved for over six months after failure of 4–5 lines of prior therapy. Both patients had high-grade localized tumors that recurred after resection with negative margins resulting in metastases to the lungs, a common site for metastatic STS [17]. This suggests the combination of nivolumab and cabozantinib has a proven safety profile and demonstrates good potential in treatment-resistant STS warranting further investigation in clinical trials. Next generation sequencing revealed that both patients had dysregulation of the cell cycle, CDKN2A/B in case 1 and TP53 in case 2, both of which are not infrequently identified with UPS. As previously mentioned, CDKN2A is a poor prognostic mutation in STS therefore it is impressive that our first patient was able to achieve disease stability for months on nivolumab/cabozantinib therapy. Other mutations noted in these two patients such as PTPN11 and HRAS have limited data in sarcomas but are associated with poor overall survival in other cancers including bladder and breast cancer [18],[19]. Interestingly, high tumor mutational burden (TMB) is often predictive of better response to immunotherapy [8]. However, both of our patients had low TMB, suggesting multiple mechanisms at play. Therefore, one could postulate that other patients with higher TMB may obtain even more favorable response from combined anti-PD1 and TKI therapy.
Tissue pathology of both patients was positive for CD99, which suggests Ewing-like sarcoma [20] but BCOR was negative by molecular testing. Given the similar cellular expression and promising results with cabozantinib in the CABONE trial where 26% of patients with Ewing sarcoma demonstrated partial response, and 33% of patients showed non-progression of disease at six months [5], it was reasonable to attempt palliative treatment with TKI therapy after multiple failed lines of therapy. The combination of PD1-inhibitor and TKI has been shown to improve overall survival in patients with metastatic renal cell carcinoma compared to TKI monotherapy. TKIs act on many receptors/pathways associated with cancer growth and development such as VEGFR2 in angiogenesis, MET in cellular growth, and many others such as RET, AXL, KIT, FLT3, etc. But over time, TKI resistance can develop from upregulation of c-MET and AXL pathway which then promotes angiogenesis and cell proliferation [21]. This results in the upregulation of PD-L1 expression which prevents T-cell mediated immune response against cancer cells [22]. The addition of nivolumab restores the immune-mediated activity against cancer cells and acts synergistically with cabozantinib to halt metastasis.
Cabozantinib is the only VEGFR2 TKI with also inhibitory effects on c-MET receptor activity [23]. In vitro studies with osteosarcoma indicate cabozantinib has a dose-dependent inhibitory effect on c-MET pathways, which downstream inhibits AKT and ERK proteins thereby inhibiting the proliferation and migration of osteosarcoma cells. Additionally, cabozantinib alters the bone microenvironment via inhibition of RANK-positive osteosarcoma cells causing a decreased amount of free RANKL, which ultimately limits the tumor cycle induced by osteoclastogenesis [24]. The anti-PD1 antibodies negatively regulate the immunomodulatory effect of tumor cells which allows them to evade the body’s immune response. In a phase 2 study, SARC028, various STS were treated with pembrolizumab monotherapy resulting in an 18% overall response with 55% of patients achieving progression-free survival at 12 weeks, and one patient with UPS even achieved complete remission [25]. A retrospective study of 88 patients with STS treated with various immune checkpoint inhibitors remarkably demonstrated that UPS had the highest rate of response [26].
Conclusion
Considering the promising results of cabozantinib and nivolumab in various tumor types and our two patients’ significant response to therapy, we conclude that further clinical trials with this combination therapy are needed in STS patients. Given the nature of case reports, it is important to highlight the inherent limitation of small patient population thereby limiting the generalizability of the findings to a broaden population. Further research, such as randomized controlled trials, is needed to validate the findings illustrated in our case report to elevate the use of combined therapy earlier in treatment guidelines to improve morbidity and mortality in aggressive STS subtypes.
REFERENCES
1.
Movva S, Wen W, Chen W, et al. Multi-platform profiling of over 2000 sarcomas: Identification of biomarkers and novel therapeutic targets. Oncotarget 2015;6(14):12234–47. [CrossRef]
[Pubmed]

2.
Survival Rates for Soft Tissue Sarcoma. American Cancer Society. 2015. [Available at: https://www.cancer.org/cancer/types/soft-tissue-sarcoma/detection-diagnosis-staging/survival-rates.html]

3.
Soft tissue sarcoma. National Comprehensive Cancer Network. 2021. [Available at: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1464]

4.
Serrano C, Romagosa C, Hernández-Losa J, et al. RAS/MAPK pathway hyperactivation determines poor prognosis in undifferentiated pleomorphic sarcomas. Cancer 2016;122(1):99–107. [CrossRef]
[Pubmed]

5.
Allen AH. Large undifferentiated pleomorphic sarcoma of the posterior thigh. Am J Case Rep 2019;20:318–22. [CrossRef]
[Pubmed]

6.
Berner K, Johannesen TB, Hall KS, Bruland ØS. Clinical epidemiology and treatment outcomes of spindle cell non-osteogenic bone sarcomas – A nationwide population-based study. J Bone Oncol 2018;14:100207.
[Pubmed]

7.
Chen S, Huang W, Luo P, et al. Undifferentiated pleomorphic sarcoma: Long-term follow-up from a large institution. Cancer Manag Res 2019;11:10001–9. [CrossRef]
[Pubmed]

8.
Zheng B, Qu Y, Wang J, Shi Y, Yan W. Pathogenic and targetable genetic alterations in resected recurrent undifferentiated pleomorphic sarcomas identified by targeted next-generation sequencing. Cancer Genomics Proteomics 2019;16(3):221–8. [CrossRef]
[Pubmed]

9.
Bui NQ, Przybyl J, Trabucco SE, et al. A clinico-genomic analysis of soft tissue sarcoma patients reveals CDKN2A deletion as a biomarker for poor prognosis. Clin Sarcoma Res 2019;9:12. [CrossRef]
[Pubmed]

10.
van der Graaf WTA, Blay JY, Chawla SP, et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2012;379(9829):1879–86. [CrossRef]
[Pubmed]

11.
Italiano A, Mir O, Mathoulin-Pelissier S, et al. Cabozantinib in patients with advanced Ewing sarcoma or osteosarcoma (CABONE): A multicentre, single-arm, phase 2 trial. Lancet Oncol 2020;21(3):446–55. [CrossRef]
[Pubmed]

12.
Ayodele O, Razak ARA. Immunotherapy in soft-tissue sarcoma. Curr Oncol 2020;27(Suppl 1):17–23. [CrossRef]
[Pubmed]

13.
Hoang NT, Acevedo LA, Mann MJ, Tolani B. A review of soft-tissue sarcomas: Translation of biological advances into treatment measures. Cancer Manag Res 2018;10:1089–114. [CrossRef]
[Pubmed]

14.
Choueiri TK, Powles T, Burotto M, et al. Nivolumab plus Cabozantinib versus Sunitinib for advanced renal-cell carcinoma. N Engl J Med 2021;384(9):829–41. [CrossRef]
[Pubmed]

15.
McGregor B, Mortazavi A, Cordes L, Salabao C, Vandlik S, Apolo AB. Management of adverse events associated with cabozantinib plus nivolumab in renal cell carcinoma: A review. Cancer Treat Rev 2022;103:102333. [CrossRef]
[Pubmed]

16.
Wunder JS, Lee MJ, Nam J, et al. Osteosarcoma and soft-tissue sarcomas with an immune infiltrate express PD-L1: Relation to clinical outcome and Th1 pathway activation. Oncoimmunology 2020;9(1):1737385. [CrossRef]
[Pubmed]

17.
Cao Y, Duan H, Su A, Xu L, Lai B. A pan-cancer analysis confirms PTPN11’s potential as a prognostic and immunological biomarker. Aging (Albany NY) 2022;14(13):5590–610. [CrossRef]
[Pubmed]

18.
Prior IA, Hood FE, Hartley JL. The frequency of RAS mutations in cancer. Cancer Res 2020;80(14):2969–74. [CrossRef]
[Pubmed]

19.
Billingsley KG, Burt ME, Jara E, et al. Pulmonary metastases from soft tissue sarcoma: Analysis of patterns of diseases and postmetastasis survival. Ann Surg 1999 May;229(5):602–10. [CrossRef]
[Pubmed]

20.
Li L, Zhang M, Chen S, et al. Detection of BCOR gene rearrangement in Ewing-like sarcoma: An important diagnostic tool. Diagn Pathol 2021;16(1):50. [CrossRef]
[Pubmed]

21.
Escudier B, Lougheed JC, Albiges L. Cabozantinib for the treatment of renal cell carcinoma. Expert Opin Pharmacother 2016;17(18):2499–504. [CrossRef]
[Pubmed]

22.
Engelsen AST, Lotsberg ML, Abou Khouzam R, et al. Dissecting the role of AXL in cancer immune escape and resistance to immune checkpoint inhibition. Front Immunol 2022;13:869676. [CrossRef]
[Pubmed]

23.
Fagioli F, Tirtei E. Cabozantinib: A new perspective for advanced bone sarcoma. Lancet Oncol 2020;21(3):331–2. [CrossRef]
[Pubmed]

24.
Fioramonti M, Fausti V, Pantano F, et al. Cabozantinib affects osteosarcoma growth through a direct effect on tumor cells and modifications in bone microenvironment. Sci Rep 2018;8(1):4177. [CrossRef]
[Pubmed]

25.
Tawbi HA, Burgess M, Bolejack V, et al. Pembrolizumab in advanced soft-tissue sarcoma and bone sarcoma (SARC028): A multicentre, two-cohort, single-arm, open-label, phase 2 trial. Lancet Oncol 2017;18(11):1493–501. [CrossRef]
[Pubmed]

26.
Monga V, Skubitz KM, Maliske S, et al. A retrospective analysis of the efficacy of immunotherapy in metastatic soft-tissue sarcomas. Cancers (Basel) 2020;12(7):1873. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
The views expressed herein are those of the author(s) and do not reflect the official policy or position of Brooke Army Medical Center, the Department of Defense, the Defense Health Agency, or any agencies under the U.S. Government.
Author ContributionsMy Linh D Vu - Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Andrew Berman - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Erica Kao - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Kristin Stoll - Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Lauren Lee - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2023 My Linh D Vu et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.