|
Case Report
A rare case of malignancy-associated ascites with no identifiable cause in a 29-year-old female with BRCA1 mutation
1 Medical student, University of Florida College of Medicine, Gainesville, Florida, United States
2 Clinical Assistant Professor, Hospital Medicine, University of Florida College of Medicine, Gainesville, Florida, United States
3 Observer, Hospital Medicine, University of Florida College of Medicine, Gainesville, Florida, United States
Address correspondence to:
Naveen Baskaran
MD, MSHI, Assistant Professor of Medicine, Division of Hospital Medicine, Department of Medicine, University of Florida, P.O. Box 100238, Gainesville, Florida 32610,
United States
Message to Corresponding Author
Article ID: 100122Z10AM2023
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Markal AC, Dideban B, Memdani A, Rathi K, Baskaran N. A rare case of malignancy-associated ascites with no identifiable cause in a 29-year-old female with BRCA1 mutation. J Case Rep Images Oncology 2023;9(1):27–31.ABSTRACT
Introduction: We present a challenging case of a 29-year-old female with a detected Breast Cancer gene 1 (BRCA1) gene mutation who presented to us with an ascites the cause of which remained undiscovered even after appropriate testing.
Case Report: A 29-year-old female presented with a five-month history of worsening intermittent abdominal pain which recently became constant and worsened in intensity without any identifiable trigger. She also had complaints of constipation for the past 2–3 days. Her past medical history was positive for (BRCA1) mutation which was tested after her mother’s diagnosis of breast cancer. Further evaluation revealed our patient had ascites, which was drained to collect 12 liters of fluid. The cytology and culture of ascitic fluid were negative, but total protein concentration or Serum to ascites albumin gradient (SAAG) ratio were concerning for a malignant cause of her ascites. She underwent multiple imaging and biopsies and the cause remain unidentified.
Conclusion: By this report we want to shed light on the diagnostic challenges of malignancy-associated ascites in patients without peritoneal carcinomatosis. The high false-negative of ascitic fluid cytology in such cases makes it an unreliable test for evaluation. We performed fresh ovarian biopsies which were benign and highlight the need for further research to assess the false negative rates in frozen versus fresh biopsies while evaluating malignancy-associated ascites in patients with an unknown primary cause.
Keywords: Ascites, Ascitic fluid analysis, BRCA-1 gene mutation, Diagnostic challenges, Diagnostic laparoscopy, Cytology of ascitic fluid, Fresh ovarian biopsy, Frozen section biopsy, Hemorrhagic cyst of the ovary, Malignancy of unknown origin, Malignancy-associated ascites, Paracentesis, Peritoneal biopsy
Introduction
While most ascites in the United States are caused by cirrhosis and portal hypertension, approximately 7% of patients with ascites develop ascites due to malignancy [1]. The most efficient way to diagnose the cause of ascites is to perform an abdominal paracentesis and subsequent ascitic fluid analysis. Depending on the profile of the ascitic fluid, such as the total protein concentration or the serum-to-ascites albumin gradient (SAAG), cytology of the ascitic fluid may be performed if the physician suspects malignancy as the underlying cause of peritoneal fluid production. However, an issue arises when a patient with malignancy-associated ascites does not have peritoneal carcinomatosis, thus increasing the false-negative rate and decreasing the sensitivity of ascitic fluid cytology. In conjunction with negative cytology, benign biopsies bring into question what other workups should be explored and whether medical management would change. We present a rare case of malignancy-associated ascites with no identified cause in a 29-year-old female with a BRCA1 pathologic mutation.
Case Report
A 29-year-old female with a past medical history significant for BRCA1 pathologic mutation presented to the emergency department with a five-month history of worsening intermittent upper abdominal pain and distension and associated constipation of two to three days duration. A few days before coming to the emergency department, the pain became constant and acutely worsened without any specific inciting event. The patient also reported associated subjective fevers, nausea, and an unintentional weight loss of six pounds since the onset of her symptoms despite the significant abdominal distension. Her last menstrual period was two days prior to her arrival at the emergency department and was unremarkable. The patient’s pain was aggravated with palpation but relieved by the pain medications she received in the emergency department. Since her mother was diagnosed with breast cancer, she underwent genetic screening in March 2020 and was found to have a BRCA1 pathologic variant; she was unable to follow up with Gynecology due to insurance coverage.
Upon arrival at the emergency department, the following labs were collected and were within normal limits: liver function tests, lipase, and B-type Natriuretic peptide (BNP); a pregnancy test was negative. An abdominal and pelvic computerized tomography (CT) showed large-volume ascites concerning for mucinous malignancy of unclear etiology without cirrhosis or organomegaly (Figure 1).
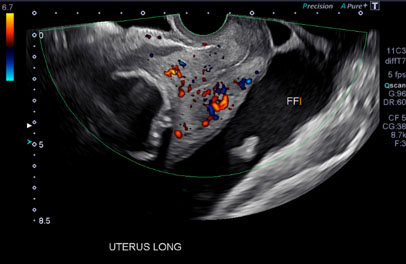
A subsequent transvaginal ultrasound showed a large volume of free fluid within the pelvis, a soft tissue nodule separated but superior to the left ovary with Doppler flow, and an enlarged uterus with a calcified fibroid (Figure 2).
Gynecologic Oncology was consulted, and this service recommended ordering the following tumor markers: Cancer antigen 125 (CA 125), Carcinoembryonic antigen (CEA), and Cancer antigen 19-9 (CA 19-9); all ordered markers were within normal limits. On the second day of admission, a paracentesis was performed, and 12 liters of brown ascites fluid were removed. Ascitic fluid cultures were performed and were negative for any pathogens. The ascitic fluid had a total protein count of 5.6 g/dL, a SAAG of 0.8 g/dL, a White blood cell count (WBC) of 500, a Polymorphonuclear leucocyte (PMN) count of less than 50%, and a Red blood cell count (RBC) of 180,000. These findings from the ascitic fluid were concerning for malignant ascites. However, the cytology of the ascitic fluid was negative. A chest CT was ordered to assess for any nodules and metastases and showed no obvious lung metastasis.
On the seventh day of the patient’s admission, magnetic resonance imaging (MRI) of the patient’s uterus and ovaries was performed. The MRI showed a lesion at the medial aspect of the right ovary, which likely represented a hemorrhagic cyst. There were no solid-enhancing ovarian or adnexal lesions concerning for primary gynecologic malignancy. The MRI also showed a large volume of ascites in the pelvis containing blood clots, more likely due to the recent paracentesis, and the aforementioned uterine fibroid.
Diagnostic laparoscopy and right ovarian cystectomy were performed on the eleventh day of admission and peritoneal biopsies were taken along with the evacuation of the ascitic fluid. Intraoperative fluid analysis showed 62,000 WBCs; 48,000 RBCs; 18% PMNs; normal levels of bilirubin and amylase; a lactate dehydrogenase (LDH) of 1,062; and a SAAG of 0.4 g/dL. The patient’s serum Lactate dehydrogenase (LDH) was 279. Quantiferon-TB Gold testing in this patient was negative. Cytology of the ascitic fluid was again negative. Pathology of the right ovarian cyst was consistent with a corpus luteum cyst, and all the surgical pathology was benign.
Discussion
In the United States, the etiology of ascites in 85% of patients with ascites stems from cirrhosis and portal hypertension [1]. Malignancy-associated ascites account for or contribute to the formation of ascites in around 7% of patients with ascites [1]. By performing an abdominal paracentesis in patients with ascites and analyzing the ascitic fluid, physicians can use this information to diagnose the cause of the ascites and determine whether the fluid is infected [2]. General ascitic fluid tests include cell count and differential, SAAG, cultures, total protein concentration, and LDH. Patients with malignancy-associated ascites, typically present with a characteristic ascitic fluid profile. In approximately 75% of patients with peritoneal carcinomatosis, the total WBC count is greater than or equal to 500 cells/mm3; having a predominance of lymphocytes indicates that the ascitic fluid instead more likely stems from malignancy rather than infection [3]. The SAAG, which identifies any existing portal hypertension, is almost always less than 1.1 g/dL in patients with peritoneal carcinomatosis [3] . The total protein concentration in patients who have malignancy-associated ascites solely due to peritoneal carcinomatosis was typically greater than or equal to 2.5 g/dL [3]. In patients who have ascites due to either malignancy or infection, the fluid-to-serum ratio is more than 1.0 as LDH is being produced or released into the ascitic fluid [3]. In 22% of patients with malignancy-associated ascites, the ascitic fluid is bloody [3].
There are a few mechanisms in which malignancy can result in the production of ascites: increased angiogenesis from tumor implants on the parietal peritoneum or omentum can lead to ascitic fluid production or tumor spread can lead to increased capillary permeability and blocked diaphragmatic lymphatics, thus also leading to ascitic fluid production [4]. Approximately two-thirds of patients with malignancy-associated ascites have peritoneal carcinomatosis, and 97% of this patient population have positive cytology of ascitic fluid [4]. Although cytology has a very high sensitivity for detecting peritoneal carcinomatosis, patients with malignancy-associated ascites solely due to massive liver metastases, malignant lymph node obstruction, or intrahepatic hepatocellular carcinoma would more likely not have positive cytology [3]. This finding is because these malignancies are less likely to have cell shedding [5]. While the specificity of ascitic fluid cytology for malignancy ranges from 97% to 100%, the sensitivity ranges from 22% to 81% with an average of 58.2% due to the lack of cell shedding in all malignancies [6],[7]. The false-negative rate of this test is reported to be as high as 42% [6]. One review found that the cytology of ascitic fluid had a sensitivity rate of 60% and a false negative rate of 19% [8]. Another study found that the sensitivity of this test was 62.4% and the false negative rate was 11.7% [7]. Motherby et al. described three factors that led to the high false-negative rate in their study: a malignancy-unrelated cause of the effusion, a screening error by the cytologist, or a sampling error by the physician [7]. Since ascitic fluid cytology has a significantly high false-negative rate, this test should not be used alone to diagnose malignancy-associated ascites.
Little research is currently available on the false negative rates of peritoneal or fresh ovarian biopsies in the setting of malignancy-associated ascites with an unknown primary tumor. Therefore, the focus will be placed on the accuracy of frozen section biopsies in diagnosing ovarian neoplasms based on the assumption that fresh biopsies, as collected in this case, are more accurate in diagnosing ovarian neoplasms. One study found that the false negative rate of frozen section biopsies in diagnosing ovarian neoplasms was 4.16% [9]. Another retrospective study found that the false negative rate was 3.5% [10]. The percentage of correct diagnoses, or accuracy, detected by both studies was 94.16% and 93.8%, respectively [9],[10]. Other studies have reported that the overall accuracy of frozen sections is 93.3% [11]. Therefore, using biopsy evaluations to identify malignant ovarian tumors have a high accuracy rate and not as significantly high false-negative rates in comparison to the cytology of ascitic fluid.
In this specific case, other possible causes of the patient’s ascites were ruled out during her admission. In patients with a SAAG of less than 1.1 g/dL, which indicates the absence of portal hypertension, other causes of ascites must be considered besides malignancy, such as peritoneal tuberculosis, pancreatitis, and nephrotic syndrome. Peritoneal tuberculosis was ruled out as the patient had a negative Quantiferon-TB Gold test. Nephrotic syndrome is unlikely in this patient as a urinalysis collected on her admission was negative for protein. Pancreatitis in this patient is also unlikely as her lipase and amylase were within normal limits and imaging showed no abnormalities of the pancreas.
In this specific case, other possible causes of the patient’s ascites were ruled out during her admission. In patients with a SAAG of less than 1.1 g/dL, which indicates the absence of portal hypertension, other causes of ascites must be considered besides malignancy, such as peritoneal tuberculosis, pancreatitis, and nephrotic syndrome. Peritoneal tuberculosis was ruled out as the patient had a negative Quantiferon-TB Gold test. Nephrotic syndrome is unlikely in this patient as a urinalysis collected on her admission was negative for protein. Pancreatitis in this patient is also unlikely as her lipase and amylase were within normal limits and imaging showed no abnormalities of the pancreas.
Conclusion
Although the cause of this patient’s malignancy-associated ascites remains unidentified, this case raises the question of what other workup should be completed, if any, in the setting of suspected malignancy-associated ascites and negative ascitic fluid cytology and benign biopsies. This case also demonstrates that relying on ascitic fluid cytology alone to diagnose malignancy-associated ascites is unreliable due to the high false-negative rates of cytology; further research needs to be conducted on the false-negative rates of peritoneal and fresh ovarian biopsies in evaluating the cause of malignancy-associated ascites.
REFERENCES
1.
Runyon BA, Montano AA, Akriviadis EA, Antillon MR, Irving MA, McHutchison JG. The serum-ascites albumin gradient is superior to the exudate-transudate concept in the differential diagnosis of ascites. Ann Intern Med 1992;117(3):215–20. [CrossRef]
[Pubmed]

2.
Runyon BA. Malignancy-related ascites and ascitic fluid “humoral tests of malignancy”. J Clin Gastroenterol 1994;18(2):94–8. [CrossRef]
[Pubmed]

3.
Runyon BA, Hoefs JC, Morgan TR. Ascitic fluid analysis in malignancy-related ascites. Hepatology 1988;8(5):1104–9. [CrossRef]
[Pubmed]

4.
Kaushik N, Khalid A, Brody D, McGrath K. EU-Sguided paracentesis for the diagnosis of malignant ascites. Gastrointest Endosc 2006;64(6):908–13. [CrossRef]
[Pubmed]

5.
Kaleta EJ, Tolan NV, Ness KA, O’Kane D, Algeciras-Schimnich A. CEA, AFP and CA 19-9 analysis in peritoneal fluid to differentiate causes of ascites formation. Clin Biochem 2013;46(9):814–8. [CrossRef]
[Pubmed]

6.
Pai RR, Shenoy KD, Minal J, Suresh PK, Chakraborti S, Lobo FD. Use of the term atypical cells in the reporting of ascitic fluid cytology: A caveat. Cytojournal 2019;16:13. [CrossRef]
[Pubmed]

7.
Motherby H, Nadjari B, Friegel P, Kohaus J, Ramp U, Böcking A. Diagnostic accuracy of effusion cytology. Diagn Cytopathol 1999;20(6):350–7. [CrossRef]
[Pubmed]

8.
Karoo ROS, Lloyd TDR, Garcea G, Redway HD, Robertson GSR. How valuable is ascitic cytology in the detection and management of malignancy? Postgrad Med J 2003;79(931):292–4. [CrossRef]
[Pubmed]

9.
da Cunha Bastos A, Salvatore CA, Faria RM. Frozen section biopsy of ovarian neoplasms. Int J Gynaecol Obstet 1983;21(2):103–10. [CrossRef]
[Pubmed]

10.
Obiakor I, Maiman M, Mittal K, Awobuluyi M, DiMaio T, Demopoulos R. The accuracy of frozen section in the diagnosis of ovarian neoplasms. Gynecol Oncol 1991;43(1):61–3. [CrossRef]
[Pubmed]

11.
Yarandi F, Eftekhar Z, Izadi-Mood N, Shojaei H. Accuracy of intraoperative frozen section in the diagnosis of ovarian tumors. Aust N Z J Obstet Gynaecol 2008;48(4):438–41. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Asena C Markal - Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Bahram Dideban - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Anisha Memdani - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Kanika Rathi - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Naveen Baskaran - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementUnfortunately, we were unable to obtain consent in this case. The diagnosis proved to be more challenging than anticipated, leading us to decide on publishing the case. Regrettably, the patient was discharged before we could secure written consent. All necessary measures have been taken to protect the patient’s identity and maintain confidentiality.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2023 Asena C Markal et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.