|
Case Report
An approach to the management of recurrent metastatic squamous cell carcinoma of the anus: A case report
1 Department of Medicine, Englewood Hospital, Englewood, New Jersey, USA
2 Student, Bergen County Academies, Hackensack, New Jersey, USA
3 Hematology Oncology Physicians of Englewood, Englewood Hospital, Englewood, New Jersey, USA
Address correspondence to:
Jason Lofters
350 Engle Street, Englewood, New Jersey,
USA
Message to Corresponding Author
Article ID: 100121Z10JL2023
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Lofters J, Elsheikh M, Mantena R, Jhawer M. An approach to the management of recurrent metastatic squamous cell carcinoma of the anus: A case report. J Case Rep Images Oncology 2023;9(1):22–26.ABSTRACT
Introduction: Anal squamous cell carcinoma (ASCC) is an uncommon gastrointestinal cancer that is increasingly curable when diagnosed in early stages. Distant metastases are relatively uncommon. New technologies have been introduced for the early detection of disease recurrence across all solid tumors, but their role in ASCC has not been well described. We present a case of isolated metachronous metastasis to the lung from primary ASCC, without primary recurrence and the application of circulating tumor DNA (ctDNA) technology in management.
Case Report: The patient is a 63-year-old female who presented with altered bowel habits, rectal bleeding and was found to have a 3×2 cm mass fixed to the anal sphincter on colonoscopy. Biopsy showed moderately differentiated invasive squamous cell carcinoma. Imaging revealed inguinal lymphadenopathy but was negative for distant metastases. She underwent definitive chemoradiotherapy without complications. Post-treatment surveillance scans were negative for residual disease or distant metastasis. Two years later, positron emission tomography (PET)/computed tomography (CT) scans revealed a 0.5 cm sub-pleural nodule which when repeated three months later grew to 0.7 cm. The nodule was then surgically resected. Histology was positive for metastatic squamous cell carcinoma, similar to the primary tumor. Given the dearth of data related to treatment in the setting of resected isolated metastatic disease we pursued ctDNA testing which was negative. Given this finding along with the clinical presentation, time to metastasis (two years) and absence of other evidence of recurrence we opted to monitor expectantly rather than administer systemic chemotherapy.
Conclusion: Anal squamous cell carcinoma with metachronous distant metastasis is an unusual phenomenon. Treatment with chemotherapy in completely resected metastatic disease is controversial. Use of ctDNA technology may have a role in aiding in this complex decision making.
Keywords: Anal squamous cell carcinoma, ctDNA, Metachronous lung metastasis
Introduction
Anal squamous cell carcinoma (ASCC) is a rare form of gastrointestinal cancer that comprises less than 1% of yearly cancer diagnoses [1]. From data collected by the American Cancer Society in 2015, there were 1010 deaths from a total of 7270 new cases of ASCC (2). Epidemiologic data suggest that, in the United States, the incidence of advanced squamous cell carcinoma tripled between 2001 and 2015, with an average annual percentage change of 8.6% in men and 7.5% in women [2]. The human papillomavirus (HPV) is a significant risk factor for ASCC, with the high risk forms (HPV-16 and HPV-18) being implicated in up to 86–97% of cases [3].
The development of distant metastases has been infrequent (10%) in patients with ASCC [3],[4],[5], with 10–20% of patients being diagnosed after being treated for locally advanced disease [5].
Patients with metastatic anal cancer generally present with synchronous metastatic disease to the lung, liver, or lymph node. The liver being the most frequent site of distant metastases [5],[6],[7]. Local tumor effects along with significant lymphadenopathy tend to contribute significantly to patient morbidity [8]. Additionally, for patients with metastatic disease a 33.5% five year relative survival rate has been reported [1]. Indicators of poor prognosis include male sex, tumor size greater than 5 cm and the presence of positive lymph nodes. Interestingly, the presence of HPV infection has been reported to be a positive prognostic marker [3],[9]. Treatment for metastatic disease typically involves combination chemotherapy/immunotherapy along with possible radiotherapy depending on the clinical scenario.
Circulating tumor DNA (ctDNA) represents tumor specific DNA mutations that can be found in the cell free component of blood. The detection of ctDNA has applications for screening, diagnosis of recurrence as well as selection of appropriate therapy [6].
In ASCC, surveillance typically involves physical examinations and imaging to identify recurrence of cancer. The concern, however, is that patients are more likely to have metastatic disease at the point of detection.
More data are now emerging about the role of ctDNA in surveillance of patients with solid organ tumors and the benefit of detection of recurrence of disease prior to clinical or radiographic evidence of recurrence [6]. Here we present a case of ASCC with isolated metachronous metastasis to the lung, without primary recurrence and how ctDNA technology was applied to help guide treatment.
Case Report
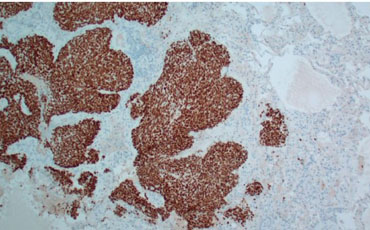
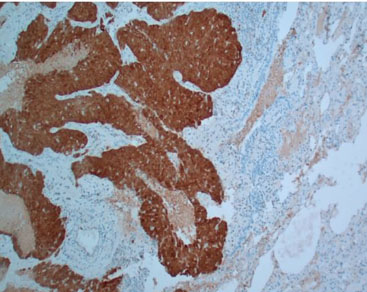
Sixty-three-year-old female who initially presented with a change in bowel habits and intermittent bright red blood per rectum. Physical exam was negative for enlarged inguinal lymph nodes. She subsequently had colonoscopy done which revealed a 1.5 cm erythematous lesion contiguous with the anal verge and encompassing 25% of its circumference. She then went on to have anoscopy and examination under anesthesia (EUA) with findings of a 3×2 cm mass in the anterior midline fixed to the sphincter complex. Biopsy of this mass revealed a moderately differentiated invasive squamous cell carcinoma with positive staining for CK5/6 and P6. The immunohistochemistry (IHC) was diffusely positive for the HPV marker P16 (Figure 1, Figure 2, Figure 3) and serum CEA at presentation was normal at 0.6 ng/mL.
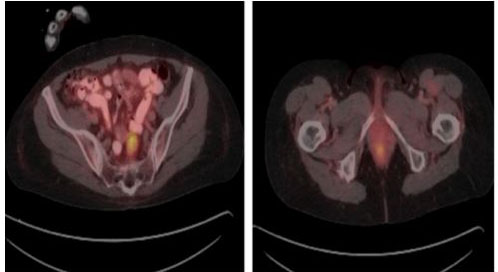
Staging computed tomography (CT) scan of the chest, abdomen, and pelvis with oral and intravenous contrast was negative for metastatic lesions but was significant for left iliac chain lymphadenopathy. A fluorodeoxyglucose positron emission tomography scan (FDG PET) was performed which demonstrated significant abnormal anal tracer uptake with SUV max 4.6 as well as tracer avid pelvic lymph nodes including presacral, medial to the left internal iliac vein tracer avid lymph nodes 2.5 1.5 cm with SUV max 6.2 as well as a left common iliac chain rounded lymph node 0.9 × 0.9 cm with SUV max 2.6, consistent with metastatic adenopathy (Figure 4).
As part of the staging evaluation, HIV testing and gynecological assessment to rule out concurrent HPV positive cervical cancer was performed and both were negative. A formal diagnosis of AJCC stage IIIA (cT2N1) anal squamous cell cancer was thus made.
She then underwent treatment with infusional 5-fluoruracil and mitomycin concurrently with external beam radiotherapy (total dose 5400 cGy). She tolerated the therapy well with no major complications.
Eight weeks after completion of chemoradiation she underwent restaging PET/CT which showed no evidence of a tracer avid anorectal lesion consistent with a complete radiographic response. As part of standard surveillance, she was expectantly monitored with physical examinations, including digital rectal examination (DRE), inguinal lymph node examination, anoscopy and carcinoembryonic antigen (CEA) every three months as well as FDGPET/CT scans every six months. A repeat colonoscopy was performed at the 1-year follow-up which revealed no polyps or anal masses, however, there was residual radiation proctitis.
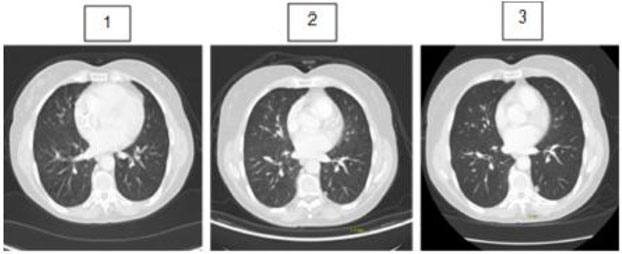
Two years after completion of concurrent chemotherapy and radiation therapy, PET/CT scans revealed a 0.5 cm left lower lobe sub-pleural nodule with SUV max 1.1 (without significant tracer uptake). She remained asymptomatic with no nausea, vomiting, altered bowel habits or change in appetite or weight. Short interval CT chest was recommended and performed three months later. It showed a significant increase in size of the nodule from 0.5 to 0.7 cm suggesting malignant potential (Figure 5). The differential diagnoses included early primary lung cancer or an isolated metastatic lesion. The case was discussed at thoracic cancer conference and a multidisciplinary decision was made for definitive surgery up front.
She underwent a left lower lobe wedge resection and mediastinal lymph node dissection. The histology of the resected lesion revealed invasive moderately differentiated squamous cell carcinoma with positive IHC staining for P63, CK5/6, and P16 and negative staining for chromogranin and synaptophysin. PDL-1 testing was positive. There was no tumor seen in the lymph nodes.
These features were in keeping with metastatic nonkeratinizing squamous cell carcinoma histologically identical to previous anal squamous cell carcinoma.
In summary, she was found to have primary anal squamous cell carcinoma with an isolated metachronous metastatic lung lesion which was surgically resected.
The dilemma was whether to treat her with adjuvant chemotherapy or not. There is a dearth of data on managing this specific condition given the rarity of ASCC. For that reason, we opted to perform ctDNA testing to better inform our decision to proceed with adjuvant chemotherapy after surgery. Testing returned negative and as such the decision was then made to pursue expectant management. To date she has remained recurrence free.
Discussion
The natural history of Anal Squamous Cell Carcinoma is somewhat indolent and the diagnosis is often delayed because common symptoms such as lower gastrointestinal bleeding, lower abdominal pain, and pressure or lower abdominal masses are often attributed to other anorectal disorders [10].
Extra-pelvic metastases are rare at the time of diagnosis and can occur in 10–20% of patients following treatment for loco-regional disease [8].
To date there have been few studies published that look specifically at the treatment of distant metastases in ASCC. Surgery has been employed in the case of solitary metastatic deposits to the brain and liver but for those with widespread metastatic disease therapy tends to involve chemotherapy/immunotherapy which is currently the recommended treatment of choice [3],[11].
In the case highlighted it is worth noting that the metastasis in this case was a metachronous one and that given its solitary nature and the initial concern for a primary early-stage lung cancer, surgery was embarked on first. Once the lesion was resected and the definitive diagnosis of metastatic ASCC was made, the dilemma faced was whether to treat with systemic chemotherapy or not.
Parallels were drawn to colon cancer patients with isolated resected metastasis. In these patients, treatment is offered in the case of isolated metastases with resection followed by six months of adjuvant chemotherapy or targeted therapy [12]. In the case of isolated metachronous lung metastasis specifically, in patients previously treated with chemotherapy there is a role for resection followed by expectant monitoring without chemotherapy [13]. To aid in the decision-making process, ctDNA technology was applied.
This technology has previously been used to aid in the identification of minimal residual disease following definitive resection in early-stage cancers as well as to the monitoring of response to systemic chemotherapy [13]. A previous study has also shown that when applied to stage III colon cancer, ctDNA has been able to successfully identify patients most at risk for recurrence after chemotherapy [14] and can thus guide treatment decisions. In the case highlighted, the decision was made to pursue expectant monitoring given the negative ctDNA results, with the view to treat with chemotherapy if same had been positive and followed by the development of new lesions clinically or on imaging.
Conclusion
Anal squamous cell carcinoma is a rarely encountered malignancy and management of metachronous metastatic disease typically involves chemotherapy. In the case of isolated metachronous disease the role of treatment with chemotherapy is less clear particularly if the lesion is successfully resected. ctDNA may aid in making that decision but additional studies are needed.
REFERENCES
1.
Surveillance, Epidemiology, and End Results (SEER) Program Research Data (1973–2015), National Cancer Institute, DCCPS, Surveillance Research Program, released April 2018, based on the November 2017 submission. Anal Cancer (1973–2009). [Available at: www.seer.cancer.gov]

2.
3.
Benson AB, Venook AP, Al-Hawary MM, et al. Anal carcinoma, version 2.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 2018;16(7):852–71. [CrossRef]
[Pubmed]

4.
Surveillance, Epidemiology, and End Results (SEER) Program SEER*Stat Database: Incidence – SEER Research Data, 8 Registries, Nov 2021 Sub (1975–2019) – Linked To County Attributes – Time Dependent (1990-2019) Income/Rurality, 1969–2020 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2022, based on the November 2021 submission. [Available at: www.seer.cancer.gov]

5.
Osborne MC, Maykel J, Johnson EK, Steele SR. Anal squamous cell carcinoma: An evolution in disease and management. World J Gastroenterol 2014;20(36):13052–9. [CrossRef]
[Pubmed]

6.
Zhang H, Lin X, Huang Y, et al. Detection methods and clinical applications of circulating tumor cells in breast cancer. Front Oncol 2021;11:652253. [CrossRef]
[Pubmed]

7.
SignateraTM. Natera. [Available at: https://www.natera.com/oncology/signatera-advanced-cancer-detection/]

8.
Eng C, Chang GJ, You YN, et al. The role of systemic chemotherapy and multidisciplinary management in improving the overall survival of patients with metastatic squamous cell carcinoma of the anal canal. Oncotarget 2014;5(22):11133–42. [CrossRef]
[Pubmed]

9.
Morton M, Melnitchouk N, Bleday R. Squamous cell carcinoma of the anal canal. Curr Probl Cancer 2018;42(5):486–92. [CrossRef]
[Pubmed]

10.
Tanum G, Tveit K, Karlsen KO, Hauer-Jensen M. Chemotherapy and radiation therapy for anal carcinoma. Survival and late morbidity. Cancer 1991;67(10):2462–6. [CrossRef]
[Pubmed]

11.
Eng C, Messick C, Glynne-Jones R. The management and prevention of anal squamous cell carcinoma. Am Soc Clin Oncol Educ Book 2019;39:216–25. [CrossRef]
[Pubmed]

12.
Valvo F, Ciurlia E, Avuzzi B, et al. Cancer of the anal region. Crit Rev Oncol Hematol 2019;135:115–27. [CrossRef]
[Pubmed]

13.
Benson AB, Venook AP, Al-Hawary MM, et al. Colon cancer, Version 2.2021, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 2021;19(3):329–59. [CrossRef]
[Pubmed]

14.
Morris VK. Circulating tumor DNA in advanced anal cancer: A blood biomarker goes viral. Clin Cancer Res 2019;25(7):2030–2. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
Mikhail Tismenetsky MD, Mathew Cherney MD
Author ContributionsJason Lofters - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Marwa Elsheikh - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Rohit Mantena - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Minaxi Jhawer - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2023 Jason Lofters et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.