|
Case Series
Radiation pneumonitis after repeat stereotactic body radiation therapy for early-stage non-small cell lung cancer: A case series of two patients
1 MD, Department of Radiology, National Hospital Organization Nagasaki Medical Center, 2-1001-1 Kubara, Omura, Nagasaki 856-0835, Japan
2 Professor, Department of Clinical Oncology, Nagasaki University Graduate School of Biomedical Sciences, 1-7-1 Sakamoto, Nagasaki 852-8501, Japan
3 MD, Department of Radiology, Sasebo City General Hospital, Hirase 9-3, Sasebo, Nagasaki 857-8511, Japan
Address correspondence to:
Kazuto Ashizawa
Department of Clinical Oncology, Nagasaki University Graduate School of Biomedical Sciences, 1-7-1 Sakamoto, Nagasaki 852-8501,
Japan
Message to Corresponding Author
Article ID: 100109Z10YT2022
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Tasaki Y, Ashizawa K, Nakamura D, Mizowaki T. Radiation pneumonitis after repeat stereotactic body radiation therapy for early-stage non-small cell lung cancer: A case series of two patients. J Case Rep Images Oncology 2022;8(2):10–14.ABSTRACT
Introduction: Stereotactic body radiation therapy (SBRT) is a well-established treatment option for patients with early-stage non-small cell lung cancer (NSCLC). We retrospectively identified 82 patients with early-stage NSCLC treated with SBRT at our institution between November 2009 and September 2019. Among these patients, two developed local recurrence or new primary lung cancer and lung metastasis or new primary lung cancer, respectively, and were treated with repeat SBRT. We herein report a case series of two patients with radiation pneumonitis after repeat SBRT.
Case Series: Case A was an 80-year-old woman diagnosed with stage I (T1aN0M0) squamous cell carcinoma. She received initial SBRT at an irradiation dose of 48 Gy in 4 fractions at the isocenter. Two years and three months after initial SBRT, the patient was clinically diagnosed with post-SBRT local recurrence or primary lung cancer and, thus, was treated with repeat SBRT at an irradiation dose of 60 Gy in 10 fractions. Six months later, the patient developed grade 5 radiation pneumonitis. Case B was an 89-year-old man diagnosed with stage I (T1cN0M0) adenocarcinoma. He received initial SBRT at an irradiation dose of 48 Gy in 4 fractions at the isocenter. Three years and six months after initial SBRT, the patient was clinically diagnosed with post-SBRT lung metastasis or primary lung cancer and, thus, was treated with repeat SBRT at an irradiation dose of 50 Gy in 4 fractions. Six months later, the patient developed grade 3 radiation pneumonitis.
Conclusion: Caution is needed when performing repeat SBRT.
Keywords: Non-small cell lung cancer, Radiation pneumonitis, Stereotactic body radiation therapy
Introduction
Stereotactic body radiation therapy (SBRT) is a well-established treatment option for patients with early-stage non-small cell lung cancer (NSCLC) [1],[2]. Excellent survival rates have been reported, especially following SBRT for early-stage NSCLC, with a high local control rate [3],[4],[5],[6]. However, a proportion of patients develop local recurrence, lung metastasis or new primary lung cancer, and treatment remains a challenge due to comorbidities that limit surgical options. Chemotherapy may be applied, but cure of the recurrent tumors may be difficult. Retreatment with conventional fractionated radiotherapy is regarded as a salvage option, but has poor outcomes [7],[8]. Since few studies have investigated repeat SBRT for reirradiation after a first course of SBRT, further information is needed on control and toxicity rates. We recently investigated the potential of repeat SBRT. We retrospectively identified 82 patients with early-stage NSCLC treated with SBRT at our institution between November 2009 and September 2019. Among these patients, two developed local recurrence or new primary lung cancer and lung metastasis or new primary lung cancer, respectively, and were treated with repeat SBRT. We herein report a case series of two patients with radiation pneumonitis after repeat SBRT, and discuss the potential of repeat SBRT considering the treatment-related toxicities.
Case Report
Case 1
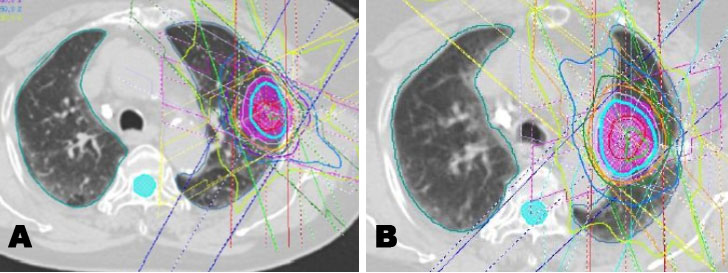
An 80-year-old woman was diagnosed with stage I (T1aN0M0) squamous cell carcinoma in left upper lobe (Figure 1A). She had a history of smoking and was being treated for liver cirrhosis. She received initial SBRT at an irradiation dose of 48 Gy in 4 fractions at the isocenter (Figure 2A). The maximal planning target volume (PTV) dose was 48 Gy. The mean lung dose (MLD) was 3.8 Gy, the volume of the lung receiving 5 Gy (lung V5) was 26%, and lung V20 was 7.5%. Follow-up examinations were conducted 1, 3, 6, 9, and 12 months after initial SBRT in the first year, and then every three months. Although the lung tumor temporarily shrank, two years and three months after initial SBRT, a new lesion, near the primary tumor, has developed in the same lobe (left upper lobe). Serial enlargement and increased uptake on fluorodeoxyglucose-positron emission tomography/computed tomography (FDG-PET/CT) were evident (Figure 1B and Figure 1C). The patient was clinically diagnosed with post-SBRT local recurrence or new primary lung cancer without pathological proof and, thus, received repeat SBRT. The repeat SBRT irradiation dose was 60 Gy in 10 fractions (Figure 2B), which was selected because local recurrence was in a central location. The maximal PTV dose was 60 Gy. Mean lung dose was 5.5 Gy, lung V5 was 20%, and lung V20 was 10%.
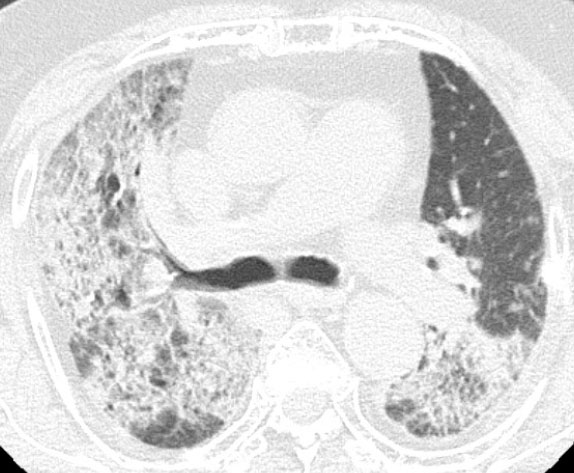
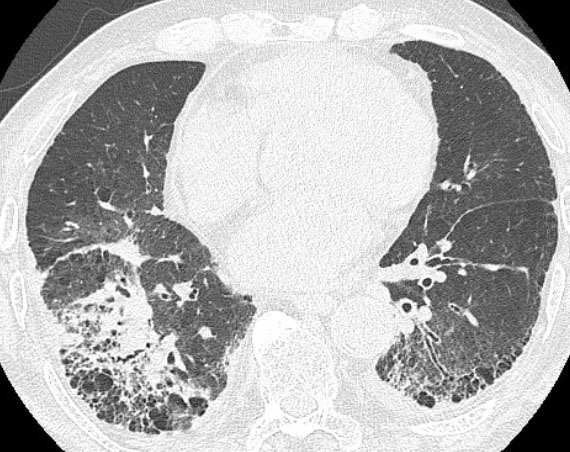
In a routine follow-up visit one and three months after end of repeat SBRT, she had no symptoms. Five months later, the patient visited the emergency room with dyspnea. Chest CT showed diffuse bilateral ground-glass opacity with interlobular septal thickening (Figure 3). High-dose methylprednisolone and empirical antibiotics were initiated for a diagnosis of radiation pneumonitis and/or pulmonary infection. Unfortunately, she failed to improve and died six months after the end of repeat SBRT. She was diagnosed with grade 5 radiation pneumonitis, which was considered to be the main cause of death.
Case 2
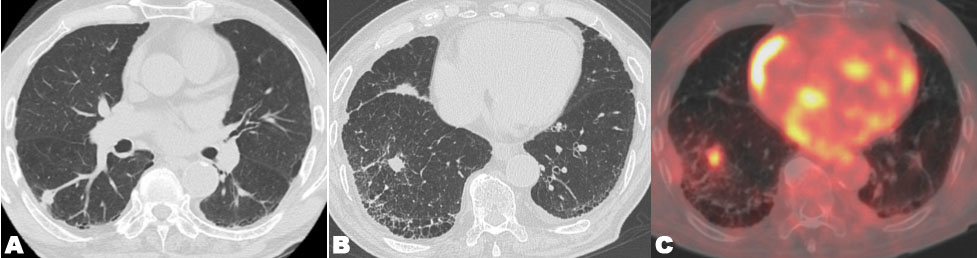
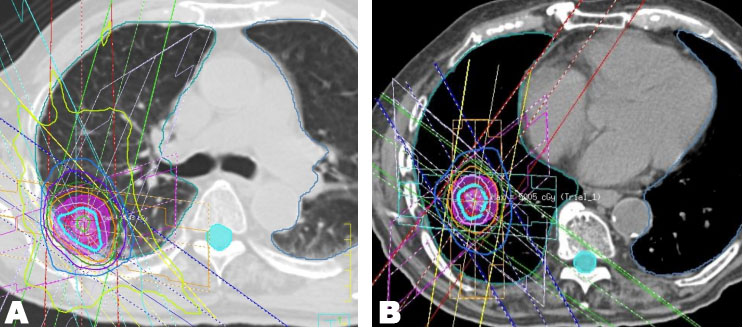
An 89-year-old man was diagnosed with stage I (T1cN0M0) adenocarcinoma in right lower lobe (Figure 4A). He had no previous history of smoking and an unremarkable medical history. He received initial SBRT at an irradiation dose of 48 Gy in 4 fractions at the isocenter (Figure 5A). The maximal PTV dose was 48 Gy. The MLD was 3.6 Gy, lung V5 was 23%, and lung V20 was 3.0%. Follow-up examinations were conducted 1, 3, 6, 9, and 12 months after initial SBRT in the first year, and then every three months. Although the lung tumor shrank, three years and six months after initial SBRT, a new lesion, distant form the lung tumor, has developed in the same lobe (right lower lobe). Serial enlargement and increased uptake on FDG-PET/CT were evident (Figure 4B and Figure 4C), which led to a diagnosis of lung metastasis or new primary lung cancer without pathological proof and, thus, the patient received repeat SBRT. The repeat SBRT irradiation dose was 50 Gy in 4 fractions (Figure 5B). The maximal PTV dose was 50 Gy. The MLD was 3.1 Gy, lung V5 was 18%, and lung V20 was 4.0%.
In a routine follow-up visit one and three months after end of repeat SBRT, he had no symptoms. In a routine follow-up visit six months after end of repeat SBRT, the patient presented with a cough. Chest CT showed consolidation with air bronchogram in the radiated field surrounded by ground-glass opacity (Figure 6). Medium-dose methylprednisolone and empirical antibiotics were initiated for a diagnosis of radiation pneumonitis and/or pulmonary infection. Since the patient required oxygen, he was diagnosed with grade 3 radiation pneumonitis. After two weeks, the attenuation of respiratory symptoms with a reduction in the patchy opacity on chest radiograph was noted. The steroid dose was tapered and discontinued.
Discussion
Increases in the risk of local recurrence or metastasis in previously irradiated lung tissue have been reported with improvements in the prognosis of patients with NSCLC [9]. Multidisciplinary teams may monitor patients or provide salvage therapy; however, the latter is often recommended because the prognosis of patients with isolated recurrence is often favorable. Reirradiation or salvage lobectomy was performed as salvage therapy. Although high local control and limited toxicity rates are associated with salvage lobectomy [10],[11], most patients with local recurrence or metastasis after SBRT are medically inoperable; therefore, the only remaining treatment option is reirradiation. Retreatment with conventional fractionated radiotherapy is regarded as a salvage option, but is associated with poor outcomes. Although repeat SBRT represents a viable option for treatment with curative intent in patients with recurrent early-stage NSCLC, it is also a significant therapeutic challenge due to life-threatening treatment-related toxicities, such as radiation pneumonitis, fatal hemorrhage after the reirradiation of centrally located tumors, and gastric perforation after the reirradiation of lower lobe tumors [12],[13].
We reported a case series of two patients with radiation pneumonitis after repeat SBRT. Patient A and patient B were diagnosed with grade 5 and 3 radiation pneumonitis, respectively. Symptomatic pneumonitis has been frequently reported after reirradiation for lung cancers [14],[15],[16]. Liu et al. [17] reported that lung V20 ≥30% of the composite plans was associated with radiation pneumonitis. Deformable image registration (DIR) was not available for generating the composite plans of our report, therefore, lung V20 of the composite plans cannot be evaluated. However, in both initial and repeat SBRT, lung V20 in patient A was higher than that in patient B, which may affect the grade of radiation pneumonitis. The incidence of radiation pneumonitis was also found to be higher after repeat SBRT for out-of-field failures than for in-field failures, and this finding was due to the exposure of large volume of normal lung tissue to low-dose radiation by treatment of out-of-field failures [18],[19]. However, in our report, patient A had an in-field failure and patient B had an out-of-field failure. The recurrent tumor in patient A was larger than the metastatic lung tumor in patient B, which may increase the lung low-dose area in patient A. In repeat SBRT, lung V5 in patient A was actually higher than that in patient B.
Previous studies demonstrated the safety of limiting the dose of repeat SBRT; however, it is important to note that an insufficient dose of fractionated reirradiation reduced the local control rate [20],[21]. Lung reirradiation studies are limited in their ability to draw definitive conclusions or guidelines regarding optimal doses, fractionation, and dose-volume constraints for safe lung retreatment. A major limitation to the interpretation of findings on repeat SBRT is the wide heterogeneity within and across studies, which includes differences between histology, recurrence patterns (local recurrence vs lung metastasis), doses, the number of fractions, radiotherapy techniques (three-dimensional non-coplanar beams vs intensity-modulated radiotherapy), the time interval between treatment courses, and whether reirradiation is performed in- or out-of-field. We believe that the irradiation dose needs to be individually and carefully selected for each case. Now we plan to introduce DIR and perform cumulative dose summation to evaluate the safety of the reirradiation.
Conclusion
Caution is needed when performing repeat SBRT.
REFERENCES
1.
Boily G, Filion É, Rakovich G, et al. Stereotactic ablative radiation therapy for the treatment of early-stage non-small-cell lung cancer: CEPO review and recommendations. J Thorac Oncol 2015;10(6):872–82. [CrossRef]
[Pubmed]

2.
Vansteenkiste J, Crinò L, Dooms C, et al. 2nd ESMO Consensus Conference on Lung Cancer: Early-stage non-small-cell lung cancer consensus on diagnosis, treatment and follow-up. Ann Oncol 2014;25(8):1462–74. [CrossRef]
[Pubmed]

3.
Temming S, Kocher M, Stoelben E, et al. Risk-adapted robotic stereotactic body radiation therapy for inoperable early-stage non-small-cell lung cancer. Strahlenther Onkol 2018;194(2):91–7. [CrossRef]
[Pubmed]

4.
Bahig H, Filion E, Vu T, et al. Excellent cancer outcomes following patient-adapted robotic lung SBRT but a case for caution in idiopathic pulmonary fibrosis. Technol Cancer Res Treat 2015;14(6):667–76. [CrossRef]
[Pubmed]

5.
Heal C, Ding W, Lamond J, et al. Definitive treatment of early-stage non-small cell lung cancer with stereotactic ablative body radiotherapy in a community cancer center setting. Front Oncol 2015;5:146. [CrossRef]
[Pubmed]

6.
Iwata H, Ishikura S, Murai T, et al. A phase I/II study on stereotactic body radiotherapy with real-time tumor tracking using CyberKnife based on the Monte Carlo algorithm for lung tumors. Int J Clin Oncol 2017;22(4):706–14. [CrossRef]
[Pubmed]

7.
Stephans KL, Woody NM, Reddy CA, et al. Tumor control and toxicity for common stereotactic body radiation therapy dose-fractionation regimens in stage I non-small cell lung cancer. Int J Radiat Oncol Biol Phys 2018;100(2):462–9. [CrossRef]
[Pubmed]

8.
Tada T, Fukuda H, Matsui K, et al. Non-small-cell lung cancer: Reirradiation for loco-regional relapse previously treated with radiation therapy. Int J Clin Oncol 2005;10(4):247–50. [CrossRef]
[Pubmed]

9.
De Ruysscher D, Faivre-Finn C, Le Pechoux C, Peeters S, Belderbos J. High-dose re-irradiation following radical radiotherapy for non-small-cell lung cancer. Lancet Oncol 2014;15(13):e620–4. [CrossRef]
[Pubmed]

10.
Antonoff MB, Correa AM, Sepesi B, et al. Salvage pulmonary resection after stereotactic body radiotherapy: A feasible and safe option for local failure in selected patients. J Thorac Cardiovasc Surg 2017;154(2):689–99. [CrossRef]
[Pubmed]

11.
Hamaji M, Chen F, Matsuo Y, Ueki N, Hiraoka M, Date H. Treatment and prognosis of isolated local relapse after stereotactic body radiotherapy for clinical stage I non-small-cell lung cancer: Importance of salvage surgery. J Thorac Oncol 2015;10(11):1616–24. [CrossRef]
[Pubmed]

12.
Nonaka H, Onishi H, Ozaki M, Kuriyama K, Komiyama T, Saito R. Serious gastric perforation after second stereotactic body radiotherapy for peripheral lung cancer that recurred after initial stereotactic body radiotherapy: A case report. J Med Case Rep 2017;11(1):343. [CrossRef]
[Pubmed]

13.
Peulen H, Karlsson K, Lindberg K, et al. Toxicity after reirradiation of pulmonary tumours with stereotactic body radiotherapy. Radiother Oncol 2011;101(2):260–6. [CrossRef]
[Pubmed]

14.
Chao HH, Berman AT, Simone CB 2nd, et al. Multiinstitutional prospective study of reirradiation with proton beam radiotherapy for locoregionally recurrent non-small cell lung cancer. J Thorac Oncol 2017;12(2):281–92. [CrossRef]
[Pubmed]

15.
Kruser TJ, McCabe BP, Mehta MP, et al. Reirradiation for locoregionally recurrent lung cancer: Outcomes in small cell and non-small cell lung carcinoma. Am J Clin Oncol 2014;37(1):70–6. [CrossRef]
[Pubmed]

16.
McAvoy S, Ciura K, Wei C, et al. Definitive reirradiation for locoregionally recurrent non-small cell lung cancer with proton beam therapy or intensity modulated radiation therapy: Predictors of high-grade toxicity and survival outcomes. Int J Radiat Oncol Biol Phys 2014;90(4):819–27. [CrossRef]
[Pubmed]

17.
Liu H, Zhang X, Vinogradskiy YY, Swisher SG, Komaki R, Chang JY. Predicting radiation pneumonitis after stereotactic ablative radiation therapy in patients previously treated with conventional thoracic radiation therapy. Int J Radiat Oncol Biol Phys 2012;84(4):1017–23. [CrossRef]
[Pubmed]

18.
Hearn JWD, Videtic GMM, Djemil T, Stephans KL. Salvage stereotactic body radiation therapy (SBRT) for local failure after primary lung SBRT. Int J Radiat Oncol Biol Phys 2014;90(2):402–6. [CrossRef]
[Pubmed]

19.
Kennedy WR, Gabani P, Nikitas J, Robinson CG, Bradley JD, Roach MC. Repeat stereotactic body radiation therapy (SBRT) for salvage of isolated local recurrence after definitive lung SBRT. Radiother Oncol 2020;142:230–5. [CrossRef]
[Pubmed]

20.
Maranzano E, Draghini L, Anselmo P, et al. Lung reirradiation with stereotactic body radiotherapy. J Radiosurg SBRT 2016;4(1):61–8.
[Pubmed]

21.
Yoshitake T, Shioyama Y, Nakamura K, et al. Definitive fractionated re-irradiation for local recurrence following stereotactic body radiotherapy for primary lung cancer. Anticancer Res 2013;33(12):5649–53.
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Yutaro Tasaki - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Kazuto Ashizawa - Conception of the work, Design of the work, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Daisuke Nakamura - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Takashi Mizowaki - Conception of the work, Design of the work, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2022 Yutaro Tasaki et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.