|
Case Report
Posterior reversible encephalopathy syndrome in a patient with hematopoietic stem cell transplant (HSCT)-associated thrombotic microangiopathy (TA-TMA)
1 DO, Resident, Internal Medicine, San Antonio Uniformed Services Health Consortium, San Antonio, TX, USA
2 MD, San Antonio Uniformed Services Health Consortium; Staff Attending, Hematology-Oncology, San Antonio, TX, USA
Address correspondence to:
Kristin E Stoll
San Antonio Uniformed Services Health Consortium, Brooke Army Medical Center, 500 Roger Brooke Drive, San Antonio, TX 78234,
USA
Message to Corresponding Author
Article ID: 100085Z10KS2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Stoll KE, Lee L. Posterior reversible encephalopathy syndrome in a patient with hematopoietic stem cell transplant (HSCT)-associated thrombotic microangiopathy (TA-TMA). J Case Rep Images Oncology 2021;7:100085Z10KS2021.ABSTRACT
Hematopoietic stem cell transplant (HSCT)-associated thrombotic microangiopathy (TA-TMA) is an increasingly recognized complication of transplantation and is associated with significant morbidity. We describe a unique case of TA-TMA associated with Streptococcus pyogenes bacteremia and posterior reversible encephalopathy syndrome (PRES) in a patient with a history of aplastic anemia who experienced complete neurologic recovery after plasma exchange and Eculizumab. This case highlights the importance of early recognition and treatment of TA-TMA and contributes to the reported cases of such occurrence.
Keywords: Posterior reversible encephalopathy syndrome; Transplant-associated thrombotic microangiopathy
Introduction
Hematopoietic stem cell transplant (HSCT)- associated thrombotic microangiopathy (TA-TMA) is an increasingly recognized complication of transplantation and is associated with significant morbidity [1]. In severe, multi-system disease, mortality is estimated between 35% and 60% [2]. In patients with TA-TMA, a concomitant diagnosis of posterior reversible encephalopathy syndrome (PRES) is rarely reported [3]. The exact pathophysiology of TA-TMA remains uncertain, but viral reactivation, graft-versus-host disease (GVHD), or systemic illness is often an inciting factor [1]. We describe a unique case of TA-TMA associated with Streptococcus pyogenes bacteremia and PRES in a patient with a history of aplastic anemia who experienced complete neurologic recovery after plasma exchange and Eculizumab.
Case Report
A 22-year-old male with very severe aplastic anemia who failed immunosuppressive therapy and was considered for therapeutic bone marrow transplant. He was without human leukocyte antigens (HLA)-matched potential donors and ultimately underwent umbilical cord stem cell transplantation following conditioning with fludarabine, cyclophosphamide, thiotepa, and total body irradiation. His course was complicated by steroid-refractory gut GVHD treated with tacrolimus, prednisone, and ruxolitinib. During treatment for GVHD he developed progressive renal dysfunction and refractory hypertension; symptoms not commonly associated with GVHD. His renal function declined precipitously prompting a renal biopsy that demonstrated findings suggestive of acute and chronic thrombotic microangiopathy (TMA), as demonstrated in Figure 1. Tacrolimus was discontinued and alternative immunosuppression was initiated. Approximately eight weeks from his original diagnosis of TMA, his course was further complicated by hypoxic respiratory failure in the setting of cryptogenic organizing pneumonia and was suspected to be pulmonary GVHD, requiring renewal of immunosuppression with increasing doses of prednisone.
During his third week of admission, in the context of prolonged immunosuppression, he subsequently developed Streptococcus pyogenes pharyngitis with rapid progression to bacteremia, for which he received clindamycin and a continuous infusion of intravenous penicillin G. He was transferred emergently to the medicine intensive care unit. Concomitant with this infection, he developed new anemia, thrombocytopenia, and severe hypertension requiring multiple agents to achieve sufficient control. Laboratory workup revealed an elevated lactate dehydrogenase (LDH), undetectable haptoglobin, negative direct antiglobulin test (DAT), elevated fibrinogen, schistocytosis, normal ADAMST13 activity, marked proteinuria, and low total hemolytic complement activity (CH50). Given his evolving clinical picture and prior renal biopsy consistent with acute and chronic changes of thrombotic microangiopathy, he was diagnosed with TA-TMA.
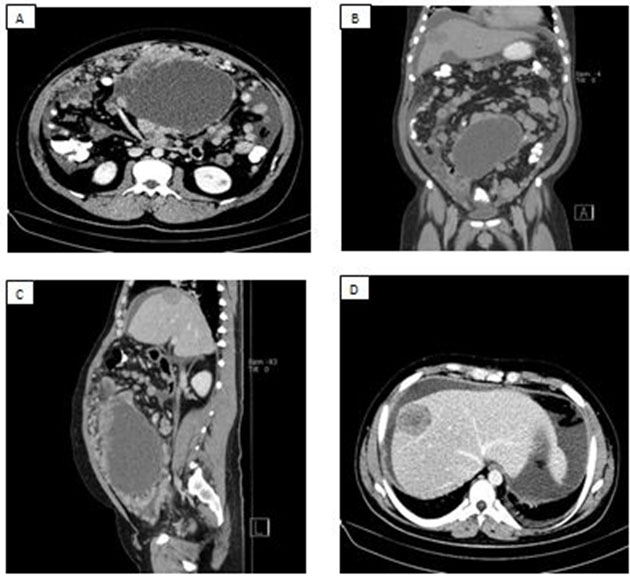
Three days after the onset of the strep pyogenes bacteremia, the patient experienced altered mental status and generalized seizure leading to pulseless electrical activity cardiac arrest. Return of spontaneous circulation was achieved following cardiopulmonary resuscitation. He remained unarousable following resuscitation. Neuroimaging revealed bilateral white matter edema in the posterior cerebral hemispheres; findings that are consistent with PRES and are depicted in Figure 2. Lumbar puncture was unremarkable without evidence of infectious process. Notable, he had no BK or JC virus reactivation. He remained hypertensive despite multiple anti-hypertensive agents and ultimately required maximal doses of a Nicardipine drip to achieve even moderate response. Electroencephalography demonstrated severe encephalopathy, but no seizure activity. Daily therapeutic plasma exchange was initiated and was given daily for three days followed by daily post-exchange Eculizumab. Remarkably, after 72 hours of therapy, he had complete neurologic recovery along with improvement in hemolysis indices. He received 10 total weeks of Eculizumab therapy.
Discussion
Posterior reversible encephalopathy syndrome is characterized by a range of neurologic symptoms including headache, visual disturbances, seizures, and other focal neurologic deficits along with distinctive neuroimaging findings demonstrating vasogenic edema. The syndrome is commonly accompanied by difficult-to-control hypertension, which is an often cited theory underlying the pathophysiology [4]. The other pathobiologic model is thought to be profound endothelial dysfunction caused by exogenous or endogenous toxins and cytokines [4]. Posterior reversible encephalopathy syndrome (PRES) is rarely reported in the literature in association with TA-TMA in the absence of calcineurin-inhibitor use. In this case, the patient had been on Tacrolimus for initial treatment of GVHD, a calcineurin-inhibitor that has been associated with PRES; however, it was discontinued four months prior to his acute diagnosis of PRES.
The diagnosis of TA-TMA is challenging as clinical symptoms and laboratory findings are non-specific and require a high level of clinical suspicion as the clinical course often mimics other common transplant associated complications, such as acute or chronic GVHD [5]. This case highlights both the challenges associated with the diagnosis and the importance of early recognition and treatment of TA-TMA, given that the treatment is drastically different from other transplants associated complications. It further contributes to the reported cases of such occurrence. In reviewing this case, particularly in the context of the thrombotic changes on renal biopsy, it is reasonable to suggest that the patient’s clinical picture was more consistent with TA-TMA rather than the initially proposed GVHD; or that this patient had concomitant and competing processes at play.
Thrombotic microangiopathy is thought to be the result of defects in the regulation of the complement system allowing for uninhibited formation of the C3 convertase C3bBb on the surface of endothelial cells. This ultimately results in the formation of the lytic complex and anaphylaxtoxin C5a leading to multi-system endothelial damage, which is the hallmark of TA-TMA [5]. At the current juncture, the most widely utilized diagnostic criteria for TA-TMA was published by Jodele et al. and utilizes histopathologic features of tissue biopsy or a diagnostic features including evidence of terminal complement activation, proteinuria, hypertension, anemia, thrombocytopenia, and evidence of microangiopathy [5].
Conclusion
In this case, widespread endothelial dysfunction likely contributed to onset of PRES. Further research is necessary to help identify methods for prevention, biomarkers for disease, and effective treatment as mortality remains high.
REFERENCES
1.
Wall SA, Zhao Q, Yearsley M, et al. Complement-mediated thrombotic microangiopathy as a link between endothelial damage and steroid-refractory GVHD. Blood Adv 2018;2(20):2619–28. [CrossRef]
[Pubmed]

2.
Dhakal P, Giri S, Pathak R, Bhatt VR. Eculizumab in transplant-associated thrombotic microangiopathy. Clin Appl Thromb Hemost 2017;23(2):175–80. [CrossRef]
[Pubmed]

3.
Bhunia N, Abu-Arja R, Bajwa RPS, Auletta JJ, Rangarajan HG. Successful treatment with eculizumab for posterior reversible encephalopathy syndrome due to underlying transplant-associated thrombotic microangiopathy in patients transplanted for sickle cell disease. Pediatr Blood Cancer 2019;66(10):e27912. [CrossRef]
[Pubmed]

4.
Fischer M, Schmutzhard E. Posterior reversible encephalopathy syndrome. J Neurol 2017;264(8):1608–16. [CrossRef]
[Pubmed]

5.
Jodele S, Laskin BL, Dandoy CE, et al. A new paradigm: Diagnosis and management of HSCT-associated thrombotic microangiopathy as multi-system endothelial injury. Blood Rev 2015;29(3):191–204. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Kristin E Stoll - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Lauren Lee - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Kristin E Stoll et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.