|
Case Report
Bilateral chylous effusions as a complication of advanced chronic lymphocytic leukemia
1 Resident, Department of Internal Medicine, Mount Sinai Medical Center, Miami Beach, FL, USA
2 Oncologist, Department of Hematology-Oncology, University of Miami Health, Miami, FL, USA
3 Pathologist, Department of Pathology, Mount Sinai Medical Center, Miami, FL, USA
4 Herbert Wertheim College of Medicine, Florida International University, Miami, FL, USA
Address correspondence to:
Megan Winter
4300 Alton Road, Miami Beach, FL,
USA
Message to Corresponding Author
Article ID: 100072Z10MW2020
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Winter M, Rodriguez E, Poppiti R. Bilateral chylous effusions as a complication of advanced chronic lymphocytic leukemia. J Case Rep Images Oncology 2020;6:100072Z10MW2020.ABSTRACT
Introduction: A chylous effusion, or chylothorax, is a collection of milky fluid consisting of emulsified fats that has accumulated in the pleural space due to extravasation from lymphatic vessels, mainly the thoracic duct. There are three main etiologies of chylothorax—traumatic, non-traumatic, and idiopathic. Non-traumatic chylothorax is typically associated with malignancy, though an association with chronic lymphocytic leukemia (CLL) is very rarely seen or reported.
Case Report: A 92-year-old woman with atrial fibrillation and CLL presented to the cancer center with the complaint of worsening shortness of breath exacerbated by exertion. Her symptoms were associated with orthopnea, paroxysmal nocturnal dyspnea, and bilateral swelling in her lower extremities. She was hospitalized and found to have a pleural effusion, believed to be due to congestive heart failure. Despite therapy with diuretics, her symptoms persisted requiring thoracentesis. It was at this time that the fluid analysis revealed a triglyceride count consistent with chylothorax. Her history was inconsistent with any common etiology for the development of chylothorax, without a history of recent surgery, recent trauma, or solid tumors. This raised suspicion that her CLL may be the cause. Less than a month later, her CLL relapsed as she exhibited worsening adenopathy and a leukocyte count that had more than doubled. She was started on steroids, venetoclax, and underwent repeat thoracentesis, ultimately requiring invasive treatment. However, she expired shortly after initiation of therapy.
Conclusion: Chronic lymphocytic leukemia has rarely been associated with the development of chylothorax for reasons that are not fully understood. It is important for clinicians to consider chylothorax in the differential diagnosis of patients with CLL presenting with shortness of breath and pleural effusion in order to guide suitable therapy, which may include adjustment of or additional chemotherapy.
Keywords: Chronic lymphocytic leukemia, Chylothorax, Chylous effusion
Introduction
Chronic lymphocytic leukemia (CLL) is a malignancy of CD5+ B cells, which leads to the accumulation of mature-appearing lymphocytes in the blood, bone marrow, and lymphoid tissues [1] The clinical course of the disease ranges from asymptomatic, requiring only monitoring, to a more aggressive disease course, requiring immediate treatment with chemotherapy. The most common complications of CLL are infection related, autoimmune related, such as autoimmune hemolysis or idiopathic thrombocytopenic purpura, and therapy related [2],[3]. Chylothorax is a very rare complication of CLL, first reported in 1990 with only 12 cases noted since that time.
Chylous pleural effusions are defined as the presence of chyle within the pleural space. The pathophysiology of this process is based on the leakage of fluid from the thoracic duct or a large communicating lymph vessel to the surrounding anatomic area. This is most often from mechanical damage during surgery, but can also be caused by obstruction to outflow of lymph, increased lymph production, or changes to the composition of lymph [4].
With a sequestration of chyle in the pleural cavity, patients also lose fat, fat-soluble vitamins, protein, electrolytes, immunoglobulins, and T-lymphocytes, which lead to malnutrition, weight loss, and an impaired immune system [5]. For a patient already suffering from immunosuppression due to malignancy, CLL in this case, this can lead to an increased risk of infection and overall mortality [6].
A chylothorax is identified by fluid analysis, which reveals a high level of triglycerides, typically higher than 110 mg/dL. Therapeutic options include conservative treatment with diet change and thoracentesis, as well as more invasive options with surgical and interventional radiology procedures [7]. In the case of a patient with CLL, though, it may provide a signal of advancing disease and an indication for further treatment with chemotherapy.
Case Report
A 92-year-old woman with history of advanced CLL and atrial fibrillation presented to the Mount Sinai Comprehensive Cancer Center in September 2018 complaining of worsening shortness of breath for two weeks. Six years prior to this presentation, the patient had been diagnosed with CLL, which was initially a Rai stage II and had progressed to advanced CLL despite continued treatment with ofatumumab, chlorambucil, and ibrutinib. Her most recent treatment regimen with ofatumumab had been started in June of 2018 after she had progression of her disease.
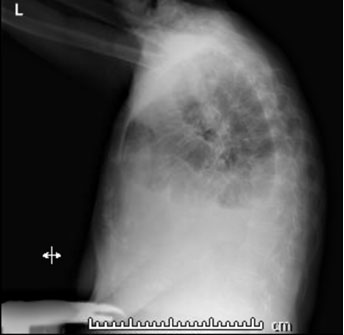
She stated that her shortness of breath was exacerbated by exertion and associated with orthopnea, paroxysmal nocturnal dyspnea, and swelling in her lower extremities. She denied any recent history of trauma, symptoms of respiratory infection, and had not had any recent surgery. After a chest X-ray was completed and demonstrated a small left and moderate right pleural effusion (Figure 1 and Figure 2), she was given a trial of furosemide. Five days later, she returned to the Cancer Center stating that her symptoms had not improved. At that time, she was noted to have a SpO2 of 85% on room air and rales in the bilateral lower lung fields. She was immediately referred to the hospital for admission.
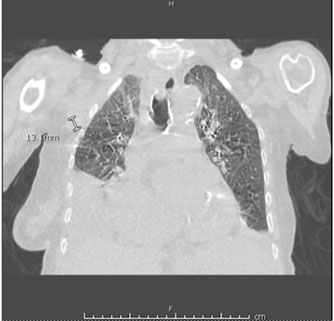
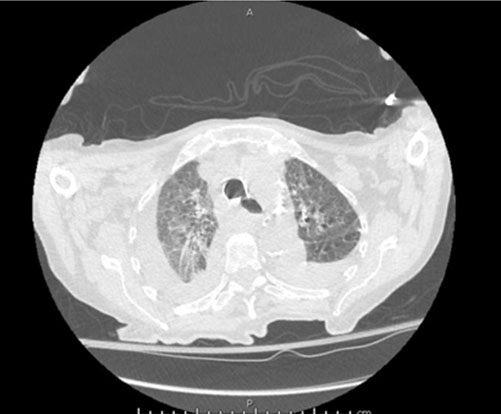
Upon arrival in the hospital, she was afebrile, normotensive, with improved O2 saturation on nasal cannula. Her physical examination was notable for atrial fibrillation, decreased breath sounds in the right lower, left middle, and left lower lung fields with accompanying rales. Her labs revealed white blood cell (WBC) of 34.25 × 103/uL with 82% lymphocytes, hemoglobin of 11.0 g/dL, platelets of 106 × 103/uL, creatinine of 1.13 mg/dL, aspartate aminotransferase (AST) of 46.0 U/L, alanine aminotransferase (ALT) of 11.0 U/L, alkaline phosphatase 120 U/L, and pro-brain natriuretic peptide (pro-BNP) 2823 pg/mL. Computed tomography of the chest (Figure 3 and Figure 4) demonstrated large right and moderate left pleural effusions with near complete and partial atelectasis with marked axillary lymphadenopathy. She was started on diuretics for suspected pleural effusion secondary to congestive heart failure.
The following morning, she underwent thoracentesis with chest tube placement to the right hemithorax. Roughly 650 cc of “milky/turbid/cloudy” fluid was drained. Analysis of the sample was consistent with an exudative fluid. The triglyceride count was 600 mg/dL, consistent with a chylous effusion. Cultures for acid-fast bacteria (AFB), fungus, and bacteria were negative. Cytology was notable for highly atypical cells suspicious for hematologic malignancy with positive staining for CD3, CD5, and CD20 showing mixed population of T and B-lymphocytes. The B-cells were positive for CD5 and CD23 consistent with CLL.
Following the procedure, the patient stated that her dyspnea had improved and she was eager to go home. She was discharged the day after thoracentesis with pleural pigtail catheter in place three days after admission. She was instructed to follow up with pulmonology and oncology and to follow a low-fat diet as to not exacerbate the production of chylous fluid.
Less than a month after discharge, her leukocyte count was noted to have increased by more than double, from 30,000 to over 70,000. In addition, she was noted to have increased adenopathy on imaging indicating a relapse of her CLL. As she had previously been treated with a Bruton tyrosine kinase inhibitor and relapsed at year six after therapy, she was started on an oral BCL2 inhibitor, venetoclax, with a planned 5-week ramp up schedule. She was monitored closely and tolerated treatment well, initially. Two weeks later, she continued to have shortness of breath and was referred for repeat thoracentesis due to recurrence of her chylothorax. She went into pulseless cardiac arrest shortly after the procedure and died in the hospital.
Discussion
A chylous effusion is the accumulation of chyle in the pleural space, manifesting as a pleural effusion with a triglyceride content of over 110 mg/dL. Chylothorax most often results in the case of thoracic surgery or tumors, with each making up about 50% of cases. The pathophysiology leading to an accumulation of chyle in the pleural space has often been attributed to a mechanical cause, one that involves damage to the lymphatic vessels resulting in a leak of fat-rich fluid. However, as more cases have been described, it seems likely that other factors such as obstruction to the lymphatic outflow, diseases of the lymph pathway affecting the integrity of the vessels, increased lymph production, and more recently described in CLL, a theory of lymph “sludging” [7]. This theory is supported by the fact that in a literature review of the prior reported cases, almost all patients had a recent history of rapid doubling in leukocyte count.
Typically, chylous effusions are treated conservatively with an emphasis on a low-fat or fat-free diet and frequent thoracentesis to remove excess fluid. If these initial measures fail, treatment with a somatostatin analog can be attempted. Surgical intervention, including thoracic duct ligation, pleurodesis, or placement of a pleuroperitoneal shunt, is the final option [7]. In the reported cases of chylothorax in patients with CLL, all patients underwent thoracentesis and required repeated drainage until other interventions were completed. One patient underwent external beam irradiation of the mediastinum with complete resolution of effusions [8]. Other patients were started on steroids and chemotherapy, most often fludarabine, though these patients expired shortly after initiating therapy [9],[10]. Our patient underwent thoracentesis on two occasions for symptomatic relief. In an effort to address the hypothesized underlying cause of worsening CLL, she also received steroids and venetoclax, the preferred therapy in someone who has progression of CLL after receiving a Bruton tyrosine kinase inhibitor. However, she only received two weeks of the BCL2 inhibitor before requiring video-assisted thoracoscopic surgery (VATS), shortly after which she expired. It is possible that her CLL, and subsequently her chylothorax, would have responded given a greater duration of therapy, though we cannot say due to her rapid expiration after VATS.
Chylothorax is a rare complication of CLL with only 12 previously reported cases. Because of this, there is little understanding as to the pathophysiology of the complication and appropriate treatment course. It is important that clinicians keep chylothorax in their differential diagnosis in patients with CLL presenting with shortness of breath in order to guide suitable therapy, which does not involve the administration of antibiotics and diuresis. One common factor among the reported cases is that the presence of a chylous effusion in a patient with CLL indicates the need for further treatment of the underlying malignancy, even in a patient with relatively stable disease.
Conclusion
Chronic lymphocytic leukemia has rarely been associated with the development of a chylothorax. Due to its rarity, the physiology behind the development and the most appropriate therapy has not been fully studied or understood. This case highlights the importance of chylothorax as a consideration in patients with CLL presenting with dyspnea and a pleural effusion. It also poses the possibility that the presence of a chylous effusion may indicate an ongoing or impending relapse of those patients previously considered in remission, triggering initiation of additional chemotherapy.
REFERENCES
1.
Kipps TJ, Stevenson FK, Wu CJ, et al. Chronic lymphocytic leukaemia. Nat Rev Dis Primers 2017;3:16096. [CrossRef]
[Pubmed]

2.
Forconi F, Moss P. Perturbation of the normal immune system in patients with CLL. Blood 2015;126(5):573–81. [CrossRef]
[Pubmed]

3.
Visco C, Barcellini W, Maura F, Neri A, Cortelezzi A, Rodeghiero F. Autoimmune cytopenias in chronic lymphocytic leukemia. Am J Hematol 2014;89(11):1055–62. [CrossRef]
[Pubmed]

4.
Doerr CH, Allen MS, Nichols FC 3rd, Ryu JH. Etiology of chylothorax in 203 patients. Mayo Clin Proc 2005;80(7):867–70. [CrossRef]
[Pubmed]

5.
Soto-Martinez M, Massie J. Chylothorax: Diagnosis and management in children. Paediatr Respir Rev 2009;10(4):199–207. [CrossRef]
[Pubmed]

6.
Breaux JR, Marks C. Chylothorax causing reversible T-cell depletion. J Trauma 1988;28(5):705–7. [CrossRef]
[Pubmed]

7.
Schild HH, Strassburg CP, Welz A, Kalff J. Treatment options in patients with chylothorax. Dtsch Arztebl Int 2013;110(48):819–26. [CrossRef]
[Pubmed]

8.
Ampil FL, Burton GV, Hardjasudarma M, Stogner SW. Chylous effusion complicating chronic lymphocytic Leukemia. Leuk Lymphoma 1993;10(6):507–10. [CrossRef]
[Pubmed]

9.
Kohmoto O, Kawabe K, Ono H, et al. Chylothorax associated with chronic lymphocytic leukemia. Intern Med 2016;55(24):3641–4. [CrossRef]
[Pubmed]

10.
Tanriverdi H, Uygar F, Tilkan OK, Gökçe M, Tor M. Chylothorax due to leukemic infiltration in a patient with chronic lymphocytic leukemia. Respir Med Case Rep 2015;16:131–3. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Estelamari Rodriguez - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Megan Winter - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Robert Poppiti - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2020 Megan Winter et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.