|
Case Report
First reported case of a synchronous occurrence of cervical carcinosarcoma and endometrial adenocarcinoma showing radiological differentiation on MRI: A case report and diagnostic challenges
1 Department of Radiology, Nagasaki University Graduate School of Biomedical Sciences, Nagasaki University School of Medicine, 1-7-1 Sakamoto, Nagasaki 852-8501, Japan
2 Department of Tumor and Diagnostic Pathology, Atomic Bomb Disease Institute, Nagasaki University, 1-12-4 Sakamoto, Nagasaki 852-8523, Japan
3 Department of Radiology, Sasebo Central Hospital, Sasebo, Nagasaki, Japan
4 Department of Radiology, Sasebo City General Hospital, Sasebo, Nagasaki, Japan
5 Department of Obstetrics and Gynecology, Nagasaki University Hospital, 1-7-1 Sakamoto, Nagasaki 852-8501, Japan
Address correspondence to:
Hirofumi Koike
Department of Radiology, Nagasaki University School of Medicine, 1-7-1 Sakamoto, Nagasaki 852-8501,
Japan
Message to Corresponding Author
Article ID: 100196Z08HK2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Koike H, Kurohama H, Nakamura T, Takenoshita S, Koga M, Oka T, Morikawa M, Harada A, Toya R. First reported case of a synchronous occurrence of cervical carcinosarcoma and endometrial adenocarcinoma showing radiological differentiation on MRI: A case report and diagnostic challenges. J Case Rep Images Obstet Gynecol 2025;11(1):35–41.ABSTRACT
Introduction: Synchronous cancer, which is when a secondary cancer occurs simultaneously or within six months of a primary cancer diagnosis, is rare for malignant female genital tract neoplasms. Despite cervical and endometrial cancers being very common gynecologic malignancies, they do not frequently occur synchronously.
Case Report: Here, we report a case of a 49-year-old female patient with a synchronous occurrence of endometrial adenocarcinoma and cervical carcinosarcoma (CCS), an extremely rare and aggressive cervical cancer subtype. Transvaginal ultrasonography showed two non-contiguous masses: one in the cervical canal and one in the uterine cavity. Magnetic resonance imaging (MRI) was then used for further examination. The two masses showed similar high signal intensity on the T2-weighted images and low apparent diffusion coefficient values. T2-weighted images suggested that they were contiguous at the cervix. However, retrospective analysis indicated that the cervical mass showed a relatively higher signal intensity on the T2-weighted images and a stronger, more heterogenous enhancement on the early-phase contrast fat-suppressed T1-weighted images. Additionally, constricted morphology was observed in the cervix, which is not consistent with what is typically observed with invasive endometrial or cervical cancer. Further analysis using histopathology and immunohistochemistry methods indicated a synchronous occurrence of CCS and endometrial adenocarcinoma, which is very rare.
Conclusion: This case effectively demonstrates the diagnostic challenges associated with magnetic resonance imaging (MRI)-based interpretation of synchronous gynecologic cancers, as well as highlights the key imaging features that may help facilitate differentiation of the two pathological types.
Keywords: Carcinosarcoma, Cervical cancer, Endometrial cancer, Magnetic resonance imaging
Introduction
Synchronous cancer is when a secondary independent cancer occurs at the same time or within six months of the primary cancer diagnosis [1]. Synchronously occurring primary malignant female genital tract neoplasms are rare [2],[3],[4],[5], with the most common types being ovary and uterus synchronous primary tumors [6]. Endometrial cancer is the second most common gynecologic cancer worldwide after cervical cancer [7]. Although cervical and endometrial cancers are very common gynecologic malignancies, they do not frequently occur as synchronous primary malignancies. Moreover, cervical carcinosarcoma (CCS) is an extremely rare, aggressive histological cervical cancer subtype. It is characterized by the presence of both epithelial and mesenchymal malignant components and accounts for less than 0.005% of all cervical cancer cases [8],[9]. The treatment approach for CCS is not well defined. However, because the stages vary depending on whether the cervical and corpus cancers are the same contiguous lesion or separate lesions, accurate diagnosis is imperative for proper treatment decisions. When conducting magnetic resonance imaging (MRI), endometrial cancer often shows a lower signal intensity compared with the normal endometrium and a higher signal intensity compared with the normal muscle layer on T2-weighted images, as well as poor enhancement on contrast fat-suppressed T1-weighted images [10],[11]. Additionally, many cervical tumors show a higher signal intensity than the cervical interstitium on T2-weighted images [12]. We chose the T2-weighted images, contrast fat-suppressed T1-weighted images, and ADC map images for cervical cancer and endometrial cancer diagnosis, as they show the characteristic findings associated with these cancers [10],[13],[14],[15],[16],[17]. Here, we report a case of a female patient with a synchronous occurrence of CCS and endometrial adenocarcinoma, which showed two similar signal masses on magnetic resonance imaging (MRI) and were difficult to diagnose. We present this case to demonstrate the diagnostic challenges associated with MRI interpretation of synchronous gynecologic cancers and to highlight the key imaging features that may facilitate differentiation.
Case Report
A 49-year-old female (body mass index, 24.2 kg/m2, gravida 3, para 3) presented with a 2-month history of irregular vaginal spotting bleeding. Her vaginal bleeding became progressively worse, resulting in secondary anemia. She visited a local hospital, where an ultrasound was performed. The ultrasound showed two masses, one in the cervix and one in the corpus of the uterus, which were suspected to be malignant tumors. She was referred to our hospital for the disease to be investigated and treated. Her medical history included anemia and her family history was unremarkable. There was no family history of gynecologic cancers. She was not taking any medication when she presented.
Upon admission at our hospital, the patient’s temperature was 36.1°C, her pulse was regular at 91 beats/minute, and her blood pressure was 136/79 mmHg.
Physical examination
A physical examination revealed a soft abdomen and palpable enlarged uterus. The pelvic examination revealed a large movable cervix with a grossly smooth appearance, well supported smooth vagina, free parametrium, and movable uterosacral ligaments.
The laboratory analysis revealed a white blood cell count of 6.0 × 109/L (normal: 3.3–9.0 × 109/L), red blood cell count of 423 × 104/µL (normal: 386–492 × 104/µL), platelet count of 315 × 103/µL (normal: 158–348 × 103/µL), hemoglobin level of 10.3 g/dL (normal: 11.6–14.8 g/dL), and C-reactive protein concentration of 0.05 mg/dL (normal concentration: ≤0.1 mg/dL). The patient had a carcinoembryonic antigen (ovarian cancer and cervical cancer biomarker) level of 0.7 ng/mL (normal: ≤5.0 ng/mL), carbohydrate antigen 125 (ovarian cancer, endometrial cancer, and cervical cancer biomarker) level of 8.7 U/mL (normal: ≤35.0 U/mL), and carbohydrate antigen 19-9 (ovarian cancer and endometrial cancer biomarker) level of 11.9 U/mL (normal: ≤ 37.0 U/mL).
Transvaginal ultrasonography
Transvaginal ultrasonography showed two masses with blood flow: one in the cervical canal (30 × 40 mm) and one in the uterine cavity (40 × 50 mm). These masses were not contiguous. The ovaries were not enlarged and no ascites was observed.
Magnetic resonance imaging (MRI)
Initial findings
Magnetic resonance imaging was performed to confirm the nature of the mass in the uterus. The lobulated and well-defined masses in the cervix and corpus of the uterus displayed similar relatively high signal intensity on the T2-weighted images (Figure 1). They also both showed a low apparent diffusion coefficient (ADC) value (Figure 2). Moreover, the masses appeared to be contiguous at the cervix on the T2-weighted images. This finding differed from what was observed using transvaginal ultrasonography.
Differential diagnosis using MRI
From the MRI results, we initially determined that the mass was uterine corpus cancer with cervical invasion or uterine cervical cancer with corpus invasion.
Retrospective analysis
Retrospectively, the cervical mass showed a relatively higher signal intensity compared with the mass in the corpus of the uterus on the T2-weighted images. The cervical mass showed a stronger and more heterogeneous enhancement compared with the mass in the corpus of the uterus on the early-phase contrast fat-suppressed T1-weighted images (Figure 3A). In addition, the late-phase contrast fat-suppressed T1-weighted images showed a slight overall enhancement in the mass of the corpus of the uterus, while they showed a relatively strong heterogeneous enhancement in the cervical mass (Figure 3B). Furthermore, the constricted morphology observed in the cervix was not considered typical for invasion of endometrial cancer or cervical cancer.
The patient underwent a cervical biopsy and fractional curettage of the endometrium. The pathological examination showed a suspected neuroendocrine carcinoma in the cervix and an adenocarcinoma in the uterine cavity. According to the International Federation of Gynecology and Obstetrics (FIGO) classification, the cervical cancer was at clinical stage Ib3 and the endometrial cancer was at stage Ia. Therefore, the patient underwent abdominal staging surgery, which included radical hysterectomy, bilateral salpingo-oophorectomy, and pelvic lymphadenectomy.
Pathological diagnosis
Macroscopic findings
Macroscopically, an ulcerative tumor (60 × 55 mm) was observed, which had replaced the whole layer of the cervix (Figure 4). In the uterine cavity, a protuberant tumor (25 × 15 mm) was present with superficial myometrial invasion (Figure 4).
Microscopic findings
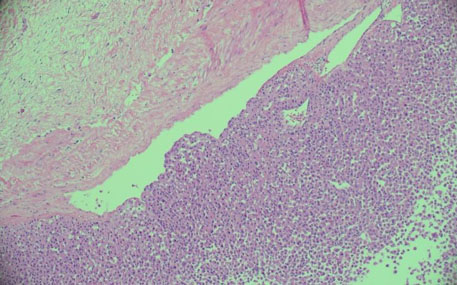
Microscopic examination showed CCS, with cervical stromal invasion (invasive depth, 17 mm), as well as endometrioid adenocarcinoma of the endometrium, grade I (Figure 5), with myometrial invasion (less than half of the entire muscle layer). Histopathology of the cervix showed highly atypical polygonal cells of various sizes that had infiltrated into the fibrous stroma.
Immunohistochemistry (IHC) results
Immunohistochemistry assays revealed positive staining for the markers AE1/AE3 (focal), vimentin (focal), and synaptophysin (focal) (Figure 6). As a result, the tumor was diagnosed as carcinosarcoma because of the presence of both epithelial and mesenchymal differentiation.
Although stromal invasion and positive lymphovascular space invasion (LVSI) were observed, no lymph node metastasis was found. The bilateral ovaries, tubes, parametrium, and vaginal surgical margins were all free. There was no LVSI of the endometrial cancer. The patient was thoroughly evaluated, with the FIGO stages determined to be cervical cancer stage Ib3 and endometrial cancer stage Ia, grade I.
The patient required postoperative chemotherapy. She underwent two courses of paclitaxel (PTX, 175 mg/m2), followed by carboplatin (area under the curve value = 6) every 3 weeks. She was undergoing follow-up at our hospital with interval clinical assessments and had no symptoms. At the time of this report, it has only been two months since her surgery. Therefore, no interval imaging studies have been performed. However, she has had no recurrence at this time.
Discussion
Synchronous gynecologic neoplasms are rare, with a reported estimated incidence of 0.7–1.8% [18]. The most common synchronous primary tumors of the genital tract are ovarian and endometrial cancers, which account for between 40% and 53% of cases [19]. Among all patients with uterine cancer, approximately 2% will be diagnosed with synchronous ovarian cancer at the time of surgery for their uterine cancer [20]. While the mechanism of this phenomenon remains unclear, it has been postulated that the embryologically similar tissues of the female genital tract may develop synchronous cancers when simultaneously subjected to irritants [3]. The most common histological subtypes of synchronous cervical and endometrial cancer include squamous cell carcinoma (SCC) of the cervix and endometrioid endometrial adenocarcinoma [21]. Cervical carcinosarcomas comprise both epithelial and mesenchymal structures and are a very rare histopathological entity, with less than 150 cases [22]. The diagnostic procedures and treatment methods of synchronous tumors do not differ from those indicated for the single primary tumors [6]. However, compared with other common cervical malignancy subtypes, such as SCC and adenocarcinoma, CCS has worse outcomes because it is prone to recurrence and metastasis [7],[22]. Surgery has been recommended as a mainstay treatment for CCS limited to the cervix, but the factors determining the necessity for and type of adjuvant therapy are not well established in this cancer type [23]. Therefore, accurate diagnosis prior to treatment is crucial.
Magnetic resonance imaging is a well-established method for diagnosing and staging uterine tumors, with T2-weighted imaging, diffusion-weighted imaging/ADC map imaging, and contrast-enhanced imaging now recommended [24],[25]. Endometrial carcinoma typically shows a lower signal intensity than the normal endometrium and higher signal intensity than the normal muscle layer on the T2-weighted images, as well as poor enhancement on the contrast fat-suppressed T1-weighted images [10],[11]. The uterine cancer in this case also showed these typical findings. Cervical carcinoma typically shows an early stronger enhancement compared with the surrounding stroma and myometrium [14]. The cervical cancer in this case also showed strong early enhancement, but the heterogeneous enhancement was somewhat atypical.
Because CCS is a very rare tumor, few comprehensive imaging studies have been published. However, Li et al. [26] reported that three cases of CCS showed a cervical mass with cystic and solid components with heterogeneous enhancement. They presented a mixed or low signal intensity on the T1-weighted images and a mixed or high signal intensity on the T2-weighted images. In our case, the MRI initially suggested that the CCS and endometrial adenocarcinoma appeared to be the same lesion. Retrospectively, the CCS showed an apparently stronger and more heterogeneous enhancement on the contrast fat-suppressed T1-weighted images, as well as a relatively higher signal intensity on the T2-weighted images and lower ADC value compared with the endometrial adenocarcinoma. It is unclear why the CCS showed a higher signal intensity on the T2-weighted images and lower ADC value. However, the CCS showed heterogeneous enhancement potentially because it contained multiple different components.
To the best of our knowledge, this is the first report of a CCS and endometrial adenocarcinoma synchronous occurrence. Furthermore, there have been no reports comparing these two lesions in the same patient using MRI. It is important to confirm when uterine and cervical cancers are occurring simultaneously. Additionally, we must be able to use MRI to determine if the lesion in the uterine body is continuous with that in the cervix. In particular, CCS may be characterized by the appearance of heterogeneous contrast enhancement. However, this case report does have certain limitations. First, the findings are limited to a single case, possibly causing generalization to be difficult. Moreover, there have been few studies regarding MRI of CCS tumors, making it unclear if the MRI findings in this case are typical. Therefore, further studies are needed.
Conclusion
Synchronous cervical cancer and endometrial cancer are rare conditions. The cervical cancer being CCS is even rarer. However, the treatment approach for multiple cancers is determined by the stage and histological type of each cancer. Therefore, clinicians should be aware that individual cancers can occur in the cervix and uterine corpus. Detailed evaluation of MRI results may be useful for predicting the pathological type of these lesions.
REFERENCES
1.
Hayat MJ, Howlader N, Reichman ME, Edwards BK. Cancer statistics, trends, and multiple primary cancer analyses from the Surveillance, Epidemiology, and End Results (SEER) Program. Oncologist 2007;12(1):20–37. [CrossRef]
[Pubmed]

2.
Axelrod JH, Fruchter R, Boyce JG. Multiple primaries among gynecologic malignancies. Gynecol Oncol 1984;18(3):359–72. [CrossRef]
[Pubmed]

3.
Eisner RF, Nieberg RK, Berek JS. Synchronous primary neoplasms of the female reproductive tract. Gynecol Oncol 1989;33(3):335–9. [CrossRef]
[Pubmed]

4.
Robboy SJ, Datto MB. Synchronous endometrial and ovarian tumors: Metastatic disease or independent primaries? Hum Pathol 2005;36(6):597–9. [CrossRef]
[Pubmed]

5.
Walsh C, Holschneider C, Hoang Y, Tieu K, Karlan B, Cass I. Coexisting ovarian malignancy in young women with endometrial cancer. Obstet Gynecol 2005;106(4):693–9. [CrossRef]
[Pubmed]

6.
Zaino R, Whitney C, Brady MF, DeGeest K, Burger RA, Buller RE. Simultaneously detected endometrial and ovarian carcinomas—A prospective clinicopathologic study of 74 cases: A gynecologic oncology group study. Gynecol Oncol 2001;83(2):355–62. [CrossRef]
[Pubmed]

7.
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010;127(12):2893–917. [CrossRef]
[Pubmed]

8.
Wright JD, Rosenblum K, Huettner PC, et al. Cervical sarcomas: An analysis of incidence and outcome. Gynecol Oncol 2005;99(2):348–51. [CrossRef]
[Pubmed]

9.
Albert A, Lee A, Allbright R, Vijayakumar S. Primary sarcoma of the cervix: An analysis of patient and tumor characteristics, treatment patterns, and outcomes. J Gynecol Oncol 2020;31(3):e25. [CrossRef]
[Pubmed]

10.
Takahashi S, Murakami T, Narumi Y, et al. Preoperative staging of endometrial carcinoma: Diagnostic effect of T2-weighted fast spin-echo MR imaging. Radiology 1998;206(2):539–47. [CrossRef]
[Pubmed]

11.
Ito K, Matsumoto T, Nakada T, Nakanishi T, Fujita N, Yamashita H. Assessing myometrial invasion by endometrial carcinoma with dynamic MRI. J Comput Assist Tomogr 1994;18(1):77–86. [CrossRef]
[Pubmed]

12.
Togashi K, Nishimura K, Itoh K, et al. Uterine cervical cancer: Assessment with high-field MR imaging. Radiology 1986;160(2):431–5. [CrossRef]
[Pubmed]

13.
Yamashita Y, Harada M, Sawada T, Takahashi M, Miyazaki K, Okamura H. Normal uterus and FIGO stage I endometrial carcinoma: Dynamic gadolinium-enhanced MR imaging. Radiology 1993;186(2):495–501. [CrossRef]
[Pubmed]

14.
Abe Y, Yamashita Y, Namimoto T, et al. Carcinoma of the uterine cervix. High-resolution turbo spin-echo MR imaging with contrast-enhanced dynamic scanning and T2-weighting. Acta Radiol 1998;39(3):322–6. [CrossRef]
[Pubmed]

15.
Woo S, Suh CH, Kim SY, Cho JY, Kim SH. Magnetic resonance imaging for detection of parametrial invasion in cervical cancer: An updated systematic review and meta-analysis of the literature between 2012 and 2016. Eur Radiol 2018;28(2):530–41. [CrossRef]
[Pubmed]

16.
Kuang F, Ren J, Zhong Q, Liyuan F, Huan Y, Chen Z. The value of apparent diffusion coefficient in the assessment of cervical cancer. Eur Radiol 2013;23(4):1050–8. [CrossRef]
[Pubmed]

17.
Andreano A, Rechichi G, Rebora P, Sironi S, Valsecchi MG, Galimberti S. MR diffusion imaging for preoperative staging of myometrial invasion in patients with endometrial cancer: A systematic review and meta-analysis. Eur Radiol 2014;24(6):1327–38. [CrossRef]
[Pubmed]

18.
Tong SY, Lee YS, Park JS, Bae SN, Lee JM, Namkoong SE. Clinical analysis of synchronous primary neoplasms of the female reproductive tract. Eur J Obstet Gynecol Reprod Biol 2008;136(1):78–82. [CrossRef]
[Pubmed]

19.
D?bska-Szmich S, Czernek U, Krakowska M, et al. Synchronous primary ovarian and endometrial cancers: A series of cases and a review of literature. Prz Menopauzalny 2014;13(1):64–9. [CrossRef]
[Pubmed]

20.
Matsuo K, Machida H, Blake EA, et al. Trends and outcomes of women with synchronous endometrial and ovarian cancer. Oncotarget 2018;9(47):28757–71. [CrossRef]
[Pubmed]

21.
Lv S, Xue X, Sui Y, et al. Synchronous primary malignant neoplasms of the cervix and endometrium. Mol Clin Oncol 2017;6(5):661–4. [CrossRef]
[Pubmed]

22.
Arend R, Doneza JA, Wright JD. Uterine carcinosarcoma. Curr Opin Oncol 2011;23(5):531–6. [CrossRef]
[Pubmed]

23.
Kimyon Comert G, Turkmen O, Karalok A, Basaran D, Bulbul D, Turan T. Therapy modalities, prognostic factors, and outcome of the primary cervical carcinosarcoma: Meta-analysis of extremely rare tumor of cervix. Int J Gynecol Cancer 2017;27(9):1957–69. [CrossRef]
[Pubmed]

24.
Nougaret S, Horta M, Sala E, et al. Endometrial Cancer MRI staging: Updated Guidelines of the European Society of Urogenital Radiology. Eur Radiol 2019;29(2):792–805. [CrossRef]
[Pubmed]

25.
Manganaro L, Lakhman Y, Bharwani N, et al. Staging, recurrence and follow-up of uterine cervical cancer using MRI: Updated Guidelines of the European Society of Urogenital Radiology after revised FIGO staging 2018. Eur Radiol 2021;31(10):7802–16. [CrossRef]
[Pubmed]

26.
Li L, Huang W, Xue K, et al. Clinical and imaging features of carcinosarcoma of the uterus and cervix. Insights Imaging 2021;12(1):142. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
We thank J. Iacona, Ph.D., from Edanz (https://jp.edanz.com/ac), for editing a draft of this manuscript.
Author ContributionsHirofumi Koike - Acquisition of data, Analysis of data, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Hirokazu Kurohama - Acquisition of data, Analysis of data, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Takaaki Nakamura - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Shintaro Takenoshita - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Miyuki Koga - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Taiga Oka - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Minoru Morikawa - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ayumi Harada - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ryo Toya - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Hirofumi Koike et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.