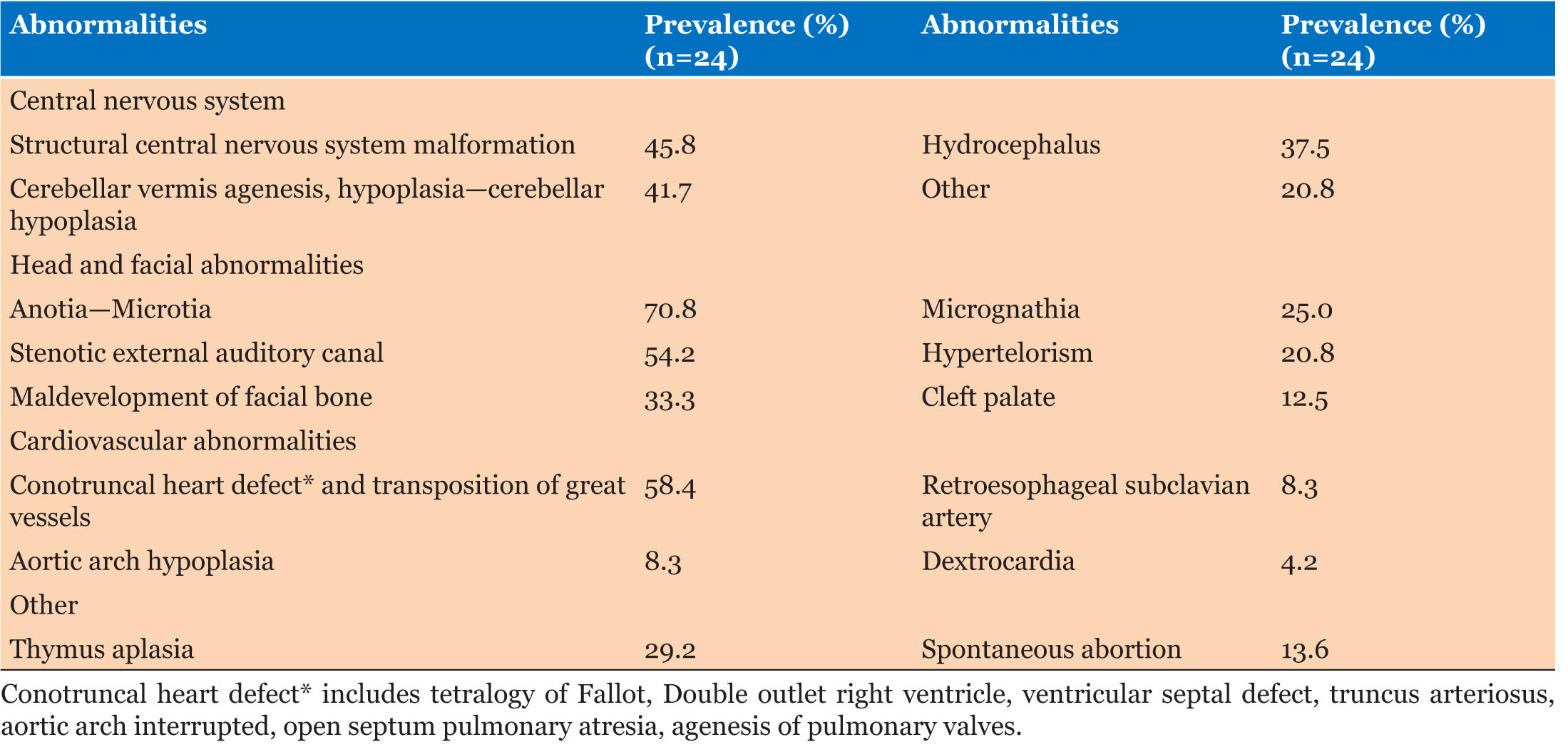
|
Case Report
A case of anti-muscle specific kinase myasthenia gravis in pregnancy with neuromuscular respiratory failure mimicking preeclampsia
1 Resident Physician, Department of Obstetrics and Gynecology, NYU Langone Hospital-Long Island, NYU Grossman Long Island School of Medicine, Mineola, NY, USA
2 Fellow, Department of Obstetrics and Gynecology, Division of Gynecologic Oncology, NYU Langone Hospital, NYU Grossman School of Medicine, New York, NY, USA
3 Assistant Professor, NYU Grossman Long Island School of Medicine, Mineola, NY, USA
4 Assistant Professor, NYU Grossman Long Island School of Medicine; Residency Program Director, Obstetrics and Gynecology, NYU Langone Hospital-Long Island, Mineola, NY, USA
5 Professor, Department of Obstetrics and Gynecology at NYU Grossman Long Island School of Medicine; Chief, Obstetrics—NYU Langone Long Island Hospital, Mineola, NY, USA
6 Chief Patient Safety Officer of Obstetrics, Northwell Health-Western Region; Professor of Obstetrics and Gynecology, Donald and Barbara Zucker School of Medicine at Hofstra/Northwell, New York, NY, USA
Address correspondence to:
Erin L Miller
MD, MPH, Department of Obstetrics and Gynecology, NYU Langone Hospital— Long Island, NYU Langone Health, 259 First Street, Mineola, NY 11501,
USA
Message to Corresponding Author
Article ID: 100191Z08EM2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Miller EL, Nawlo J, Rekawek P, Vahanian SA, Sicuranza G, Vintzileos AM. A case of anti-muscle specific kinase myasthenia gravis in pregnancy with neuromuscular respiratory failure mimicking preeclampsia. J Case Rep Images Obstet Gynecol 2025;11(1):6–10.ABSTRACT
Introduction: This report discusses a rare case of antimuscle specific kinase (MuSK) positive myasthenia gravis (MG) during pregnancy, presenting with respiratory failure mimicking preeclampsia with severe features.
Case Report: A 29-year-old primigravida presented to triage at 36 weeks with symptoms of dyspnea, orthopnea, and tachypnea necessitating cesarean delivery. Postpartum she developed neuromuscular respiratory failure leading to multiple re-intubations. Multi-system testing ultimately revealed anti-muscle specific kinase (anti-MuSK) positive serotype.
Conclusion: Despite the challenges in diagnosis and management, interdisciplinary collaboration and long-term follow-up were emphasized. Teaching points highlight the need to consider autoimmune disorders in peripartum cardiopulmonary exacerbations. The case prompts further investigation into the physiologic implications of concurrent new-onset myasthenia gravis and preeclampsia.
Keywords: Autoimmune, High risk pregnancy, Neuromuscular junction disorders, Preeclampsia
Introduction
Few cases are reported of anti-muscle specific kinase (MuSK) positive myasthenia gravis diagnosed or managed in pregnancy. In our case, this rare subtype which is commonly associated with bulbar symptoms and poor response to medical management had an unusual phenotypic manifestation of neuromuscular respiratory failure in the third trimester mimicking preeclampsia with severe features. The clinical presentation, of a condition that did not improve with delivery and subsequently worsened postpartum requiring multiple re-intubations, led to seeking a broader differential.
Case Report
A 29-year-old G4P0030 patient of Haitian descent with no medical history presented to Labor and Delivery triage at 36 weeks gestation with worsening orthopnea, dyspnea, hypoxia, and tachypnea. Her presenting vital signs were within normal limits (blood pressure 127/65 mmHg, heart rate 79 bpm), and she was saturating >95% on room air at rest. On physical examination, she had diminished bilateral breath sounds and mild lower extremity edema. During the evaluation, she required oxygen supplementation due to desaturation to 85% with exertion. Initial workup was negative for anemia, cardiac pathology, viral illness, and pulmonary embolism. Her preeclampsia labs were normal with no proteinuria. Chest X-ray revealed trace bilateral pleural effusions and poor lung volumes (Figure 1). Transthoracic echocardiogram was unremarkable with preserved ejection fraction (EF 65%) and computed tomography (CT) angiogram did not show evidence of pulmonary embolism. COVID-19 polymerase chain reaction (PCR) was negative. Laryngoscopy was performed to evaluate the upper airway, which was unremarkable. The fetal growth ultrasound one week prior to admission demonstrated appropriate fetal growth (23rd percentile). On day of admission, the biophysical profile was reassuring (8/8) and the amniotic fluid volume was normal. Due to concern for respiratory volume overload and inclusion of atypical preeclampsia with severe features in the differential, the patient received seizure prophylaxis with levetiracetam in lieu of magnesium sulfate and diuresis with furosemide. Despite these interventions, the patient’s condition deteriorated, with worsening orthopnea, tachypnea, and increasing oxygen requirement. Due to the worsening respiratory status and fetal concerns, the patient underwent primary cesarean delivery under general anesthesia and remained intubated with transfer to the surgical intensive care unit. Primary care of the patient was under the surgical intensive care unit (SICU) team with maternal–fetal medicine consultation.
On postoperative day 1, the patient was extubated. Following extubation, the patient was noted to be dyspneic with increased work of breathing requiring bilevel positive airway pressure (BiPAP) and subsequent reintubation. Further evaluation revealed mild range hypertension and transaminitis [aspartate aminotransferase (AST) 78, alanine aminotransferase (ALT) 65], now consistent with preeclampsia with severe features. On postoperative day 2, a nicardipine drip was initiated given severe range hypertension which continued until postoperative day 5. The patient remained intubated however developed abdominal distention with abdominal X-ray revealing an ileus (Figure 2). Workup by the gastroenterology team revealed colonic pseudo-obstruction requiring nasogastric tube and rectal tube placement for decompression. By postoperative day 4, the patient was again extubated, but within 12 hours, she developed significant work of breathing requiring a third intubation. Rheumatology and neurology consultations were obtained, and preliminary workup, including initial acetylcholine receptor antibodies, returned negative. By postoperative day 6, the patient developed progressive neck and bulbar muscle weakness with head droop to the right side and drooling. A discussion occurred among the care teams regarding the propensity for autoimmune diseases to present or flare in the postpartum period. Given concern for myasthenia gravis, a pyridostigmine challenge was initiated with 60 mg three times daily for five days and the patient was able to transition to continuous positive airway pressure (CPAP). With limited improvement in muscle and respiratory weakness, the pyridostigmine was discontinued on postoperative day 12. By hospital day 15, muscle-specific kinase antibody resulted positive which confirmed a diagnosis of myasthenia gravis thus pyridostigmine was restarted and prednisone was initiated. The patient’s respiratory status and strength improved with resolution of ileus. She was discharged home on hospital day 21 with a tracheostomy collar.
The neonate’s course was unremarkable. The APGAR scores were 8 at 1 minute and 9 at 5 minutes after delivery. Birth weight was 2215 g and length was 45.7 cm. The infant did not develop any muscle weakness or respiratory difficulties during the hospital stay. The infant had a prolonged stay due to maternal hospitalization but was discharged home on hospital day 10 without complication. Myasthenia gravis autoantibodies were never tested in the neonate given delay in definitive maternal diagnosis. The neonate was followed for two months after delivery with no known residual symptoms.
Discussion
Myasthenia gravis (MG) is an autoimmune disorder where preformed antibodies attack the nicotinic acetylcholine receptors at the neuromuscular junction causing skeletal muscle weakness [1],[2]. Around 75–80% of MG patients have anti-acetylcholine receptor antibodies (anti-AChR) on serology. About 40% of MG patients with seronegative MG (SNMG) have antibodies directed against muscle specific kinase (MuSK) [3],[4]. This subtype of anti-MuSK+ MG constitutes about 7–10% of all MG cases and has differences in the clinical phenotype namely, a marked female preponderance with higher incidence among African–American patients and patients living closer to the equator [5],[6],[7]. Given the increased understanding of MuSK+ MG within the last two decades, a critical feature attributed to this MG subtype is the poorer response to therapeutic regimens, with more pronounced involvement of bulbar and respiratory weakness, and lower rates of complete disease remission [8],[9],[10].
This patient’s dyspnea, initially attributed to preeclampsia, persisted post-delivery. Elevated liver enzymes, twice the normal limit, appeared within 48 hours after delivery, consistent with preeclampsia. However, multiple re-intubations postpartum demanded a broader assessment. Myasthenia gravis commonly affects patients in the second and third decades of life, which encompasses early childbearing years [11],[12],[13]. Few cases of seronegative anti-MuSK+ MG have been reported in pregnancy [14],[15],[16],[17],[18],[19],[20],[21],[22],[23]. This patient’s anti-MuSK+ MG manifested postoperatively, which deviates from the usual occurrence in the first trimester and one month postpartum [24],[25]. The case’s novelty extends beyond the MG subtype presentation. The delayed diagnosis is attributed to overlapping symptoms with postpartum preeclampsia with severe features. Few reports depict concurrent MG and preeclampsia, and even fewer show their onset in late pregnancy [26],[27],[28],[29],[30],[31],[32],[33],[34],[35],[36].
The concurrence of these two diseases is likely not by chance. Consistent with Dr. Basky Thilaganathan and others who have elucidated the connection between cardiovascular dysfunction and preeclampsia development, this patient’s autoimmune disorder may have impacted placental perfusion weeks before her myasthenia gravis symptoms [37]. Preeclampsia could be a secondary outcome. Managing these two diseases concurrently posed diagnostic and treatment challenges. This patient was placed on levetiracetam due to concern for pulmonary edema. However, this was fortuitous as magnesium sulfate, the drug of choice for seizure prophylaxis in preeclampsia, is contraindicated in myasthenia gravis due to exacerbation of neuromuscular blockade of calcium channels that can worsen a myasthenic crisis [31].
Between 10% and 20% of neonates born MG mothers develop neonatal MG due to transfer of maternal autoantibodies [38]. Although data is limited, mothers with anti-MuSK antibody-positive MG may have a higher neonatal MG risk which can cause earlier and more severe neonatal manifestations [22],[23],[37],[38]. This patient’s neonate exhibited no signs of MG after delivery. Monitoring for two months ultimately revealed no deficits. This passage of autoantibodies has been theorized to contribute to risk of polyhydramnios and fetal arthrogryposis [19],[39]. In this case, no signs of polyhydramnios or fetal arthrogryposis were observed which may be due to expedited delivery at symptoms onset.
Conclusion
A high degree of suspicion of an autoimmune etiology should be considered for any rapid onset cardiopulmonary exacerbation given the vulnerability for flares of autoimmune diseases in the peripartum period. In such cases, the presence of respiratory failure requiring repeated intubations should raise suspicion for MuSK-MG in pregnancy, especially in the presence of bulbar muscle involvement. The concurrence of new onset myasthenia gravis with preeclampsia may have deeper physiologic implications of a synergistic disease process that requires further investigation. In addition, considering the high risk of transient neonatal myasthenia gravis, close surveillance of the neonate for symptoms of respiratory distress or muscle weakness is warranted up to one week postpartum.
REFERENCES
1.
Gilhus NE. Myasthenia gravis. N Engl J Med 2016;375(26):2570–81. [CrossRef]
[Pubmed]

2.
Jayam Trouth A, Dabi A, Solieman N, Kurukumbi M, Kalyanam J. Myasthenia gravis: A review. Autoimmune Dis 2012;2012:874680. [CrossRef]
[Pubmed]

3.
Eymard B. Antibodies in myasthenia gravis. [Article in French]. Rev Neurol (Paris) 2009;165(2):137–43. [CrossRef]
[Pubmed]

4.
Hoch W, McConville J, Helms S, Newsom-Davis J, Melms A, Vincent A. Auto-antibodies to the receptor tyrosine kinase MuSK in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat Med 2001;7(3):365–8. [CrossRef]
[Pubmed]

5.
Padua L, Tonali P, Aprile I, Caliandro P, Bartoccioni E, Evoli A. Seronegative myasthenia gravis: Comparison of neurophysiological picture in MuSK+ and MuSK–patients. Eur J Neurol 2006;13(3):273–6. [CrossRef]
[Pubmed]

6.
Sanders DB, Juel VC. MuSK-antibody positive myasthenia gravis: Questions from the clinic. J Neuroimmunol 2008;201–202:85–9. [CrossRef]
[Pubmed]

7.
Deymeer F, Gungor-Tuncer O, Yilmaz V, et al. Clinical comparison of anti-MuSK- vs anti-AChR-positive and seronegative myasthenia gravis. Neurology 2007;68(8):609–11. [CrossRef]
[Pubmed]

8.
Sanadze AG, Sidnev DV, Karganov MIu. Clinical features of myasthenia related with autoantibodies to muscle specific tyrosine kinase (MuSK). [Article in Russian]. Zh Nevrol Psikhiatr Im S S Korsakova 2010;110(12):72–6.
[Pubmed]

9.
Alboini PE, Evoli A, Damato V, Iorio R, Bartoccioni E. Remission of myasthenia gravis with MuSK antibodies during ruxolitinib treatment. Muscle Nerve 2017;55(3):E12–3. [CrossRef]
[Pubmed]

10.
Huang Q, Li F, Zhao S. Spotlight on MuSK positive myasthenia gravis: Clinical characteristics, treatment and outcomes. BMC Neurol 2022;22(1):73. [CrossRef]
[Pubmed]

11.
Ferrero S, Esposito F, Biamonti M, Bentivoglio G, Ragni N. Myasthenia gravis during pregnancy. Expert Rev Neurother 2008;8(6):979–88. [CrossRef]
[Pubmed]

12.
Burke ME. Myasthenia gravis and pregnancy. J Perinat Neonatal Nurs 1993;7(1):11–21. [CrossRef]
[Pubmed]

13.
Hamel J, Ciafaloni E. An update: Myasthenia gravis and pregnancy. Neurol Clin 2018;36(2):355–65. [CrossRef]
[Pubmed]

14.
Neves AR, Monteiro P, Matos A, Santos Silva I. Anti-MuSK-positive myasthenia gravis diagnosed during pregnancy: New challenges for an old disease? BMJ Case Rep 2015;2015:bcr2014207708. [CrossRef]
[Pubmed]

15.
Terrero A, Ramírez-Rivera J. “Seronegative” anti-MUSK positive myasthenia gravis presenting during pregnancy. Bol Asoc Med P R 2006;98(3):210–2.
[Pubmed]

16.
Béhin A, Mayer M, Kassis-Makhoul B, et al. Severe neonatal myasthenia due to maternal anti-MuSK antibodies. Neuromuscul Disord 2008;18(6):443–6. [CrossRef]
[Pubmed]

17.
18.
Niks EH, Verrips A, Semmekrot BA, et al. A transient neonatal myasthenic syndrome with anti-musk antibodies. Neurology 2008;70(14):1215–6. [CrossRef]
[Pubmed]

19.
O'carroll P, Bertorini TE, Jacob G, Mitchell CW, Graff J. Transient neonatal myasthenia gravis in a baby born to a mother with new-onset anti-MuSK-mediated myasthenia gravis. J Clin Neuromuscul Dis 2009;11(2):69–71. [CrossRef]
[Pubmed]

20.
Kanzaki A, Motomura M. A pregnant patient with anti-MuSK antibody positive myasthenia gravis and her infant with transient neonatal myasthenia gravis. [Article in Japanese]. Rinsho Shinkeigaku 2011;51(3):188–91. [CrossRef]
[Pubmed]

21.
Lee JY, Min JH, Han SH, Han J. Transient neonatal myasthenia gravis due to a mother with ocular onset of anti-muscle specific kinase myasthenia gravis. Neuromuscul Disord 2017;27(7):655–7. [CrossRef]
[Pubmed]

22.
Takahashi Y, Yamashita T, Morihara R, et al. Emergency caesarean section saved both an anti-musk antibody-positive myasthenia gravis mother with pregnancy-induced hypertension and her premature baby. Intern Med 2017;56(24):3361–4. [CrossRef]
[Pubmed]

23.
Inoue KI, Tsugawa J, Fukae J, et al. Myasthenia gravis with anti-muscle-specific tyrosine kinase antibody during pregnancy and risk of neonatal myasthenia gravis: A case report and review of the literature. Case Rep Neurol 2020;12(1):114–20. [CrossRef]
[Pubmed]

24.
Téllez-Zenteno JF, Hernández-Ronquillo L, Salinas V, Estanol B, da Silva O. Myasthenia gravis and pregnancy: Clinical implications and neonatal outcome. BMC Musculoskelet Disord 2004;5:42. [CrossRef]
[Pubmed]

25.
Massey JM, De Jesus-Acosta C. Pregnancy and myasthenia gravis. Continuum (Minneap Minn) 2014;20(1 Neurology of Pregnancy):115–27. [CrossRef]
[Pubmed]

26.
Cohen BA, London RS, Goldstein PJ. Myasthenia gravis and preeclampsia. Obstet Gynecol 1976;48(1 Suppl):35S–7S.
[Pubmed]

27.
Duff GB. Preeclampsia and the patient with myasthenia gravis. Obstet Gynecol 1979;54(3):355–8.
[Pubmed]

28.
Brogan M, Corcoran DJ. Myasthenia gravis and pre-eclampsia. Ir Med J 1983;76(2):84–5.
[Pubmed]

29.
Benshushan A, Rojansky N, Weinstein D. Myasthenia gravis and preeclampsia. Isr J Med Sci 1994;30(3):229–33.
[Pubmed]

30.
Hamaoui A, Mercado R. Association of preeclampsia and myasthenia: A case report. J Reprod Med 2009;54(9):587–90.
[Pubmed]

31.
Di Spiezio Sardo A, Taylor A, Pellicano M, et al. Myasthenia and HELLP syndrome. Eur J Obstet Gynecol Reprod Biol 2004;116(1):108–11. [CrossRef]
[Pubmed]

32.
Hassan A. Myasthenia gravis and preeclampsia: Dot all the I’s and cross all the T’s. J Taibah Univ Med Sci 2017;12(5):461–4. [CrossRef]
[Pubmed]

33.
Sikka P, Joshi B, Aggarwal N, Suri V, Bhagat H. Distinguishing myasthenia exacerbation from severe preeclampsia: A diagnostic and therapeutic challenge. J Clin Diagn Res 2015;9(8):QD05–6. [CrossRef]
[Pubmed]

34.
Mueksch JN, Stevens WA. Undiagnosed myasthenia gravis masquerading as eclampsia. Int J Obstet Anesth 2007;16(4):379–82. [CrossRef]
[Pubmed]

35.
Lake AJ, Al Khabbaz A, Keeney R. Severe preeclampsia in the setting of myasthenia gravis. Case Rep Obstet Gynecol 2017;2017:9204930. [CrossRef]
[Pubmed]

36.
Ozcan J, Balson IF, Dennis AT. New diagnosis myasthenia gravis and preeclampsia in late pregnancy. BMJ Case Rep 2015;2015:bcr2014208323. [CrossRef]
[Pubmed]

37.
Thilaganathan B. The placenta is the villain or victim in the pathogenesis of pre-eclampsia. BJOG 2021;128(2):148. [CrossRef]

38.
Iijima S. Clinical and pathophysiologic relevance of autoantibodies in neonatal myasthenia gravis. Pediatr Neonatol 2021;62(6):581–90. [CrossRef]
[Pubmed]

39.
Piccioni MG, Tabacco S, Giannini A, Deroma M, Logoteta A, Monti M. Myasthaenia gravis in pregnancy, delivery and newborn. Minerva Ginecol 2020;72(1):30–5. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Erin L Miller - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
J Nawlo - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
P Rekawek - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
SA Vahanian - Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
G Sicuranza - Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
AM Vintzileos - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Erin L Miller et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.