|
Case Report
Abdominal aortitis with immune checkpoint inhibition
1 Lewis Katz School of Medicine, Temple University, Philadelphia, PA, USA
2 Lifespan Cancer Institute, Rhode Island Hospital, Providence, RI, USA
3 Warren Alpert School of Medicine, Brown University, Providence, RI, USA
Address correspondence to:
Nicholas R Liguori
Lewis Katz School of Medicine, Temple University, Philadelphia, PA,
USA
Message to Corresponding Author
Article ID: 100092Z10NL2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Liguori NR, Longo MV, Safran H. Abdominal aortitis with immune checkpoint inhibition. J Case Rep Images Oncology 2021;7:100092Z10NL2021.ABSTRACT
Introduction: Aortitis, inflammation of the aorta, is a rare large vessel vasculitis associated with giant cell arteritis, Takayasu arteritis, and other rheumatologic diseases. Aortitis has rarely been reported with cancer immunotherapy.
Case Report: We present a case of 43-year-old man treated for a high-grade neuroendocrine tumor who developed aortitis during treatment with the PD-L1 inhibitor atezolizumab. PubMed search revealed no prior cases of this toxicity in associated with PD-L1 inhibition.
Conclusion: To our knowledge this is the first case report of aortitis from PD-L1 inhibition. Recognition is important since resolution of symptoms was obtained with corticosteroids. This is also the first report of successful rechallenge with a PD-1 inhibitor without recurrence of aortitis.
Keywords: Aortitis, Atezolizumab, PD-1 inhibitor, PDL1 inhibitor
Introduction
Increasing tumor-specific T-cell immunity by inhibiting programmed death ligand 1 (PD-L1) and programmed death 1 (PD-1) signaling has shown promise in the treatment of non-small cell and small cell lung cancer, melanoma, kidney cancer, hepatocellular cancer, and other malignancies [1]. While there are substantial potential clinical benefits to immune checkpoint inhibition, there is a spectrum of toxicities termed immune-related adverse events (irAEs) [1]. Virtually any organ system in the body may be affected. The most common irAEs are pulmonary, dermatologic, gastrointestinal, hepatic and endocrine. While uncommon, fatal toxicities may develop following immune checkpoint inhibition and prompt recognition is important
Some patients with significant irAEs during initial treatment with a PD-1/PD-L1 checkpoint inhibitor may be retreated. The choice to retreat is dependent on multiple factors, including the severity and nature of the initial irAE and success of resolution of toxicity with immunosuppression. The clinical course of a patient with aortitis, amelioration of toxicity with corticosteroids, and rechallenge with immunotherapy is described.
Case Report
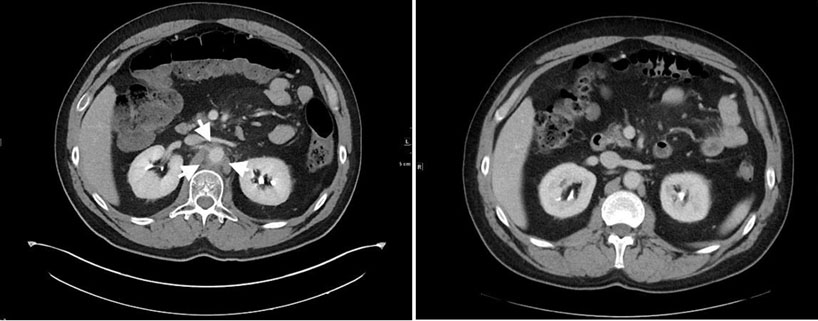
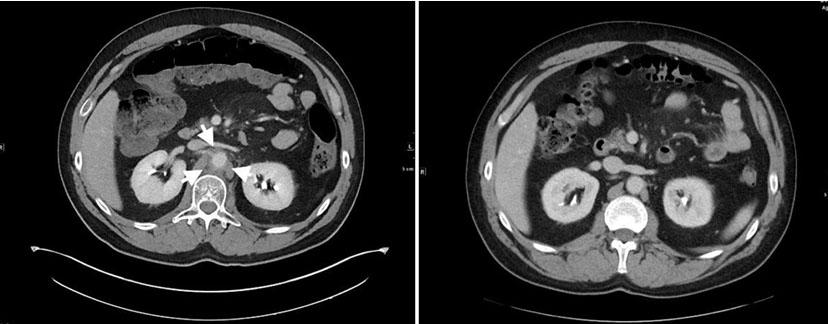
This is a man who presented in April 2017 with left upper quadrant abdominal pain. Computed tomography (CT) of the abdomen showed a mass in the tail of the pancreas and diffuse liver metastases. Pathology from the liver biopsy showed a high-grade neuroendocrine cancer with numerous mitotic figures and apoptotic bodies, and Ki-67 proliferation index of 90%. Chest CT was negative. The patient’s tumor underwent genomic analysis in May 2017 utilizing FoundationOne, a next-generation sequencing (NGS) based assay. The assay performed on his sample interrogated 315 genes as well as introns of 28 genes involved in rearrangements. Genomic alternations identified in his sample included RB1 loss exons 18–27 and GABRA6 V2281. The patient received 7 cycles of carboplatin and etoposide and obtained a complete radiographic response. In October 2018 he developed a recurrence in the liver and was retreated with carboplatin and etoposide with the addition of the PD-L1 inhibitor atezolizumab. After two cycles of therapy, administered at 21-day intervals, he developed abdominal pain. Computed tomography scan showed inflammation and stranding around the aorta consistent with aortitis. Prednisone was initiated, 1 mg/kg and the pain resolved within a week. He continued treatment on chemotherapy alone while receiving a 2-month prednisone taper. After 6 cycles of chemotherapy, he again achieved a remission. Three months after discontinuation of therapy he recurred with diffuse liver metastases. Atezolizumab was reinitiated with carboplatin and etoposide. After two cycles, he again developed severe abdominal pain. Computed tomography scan (Figure 1) showed interval development of significant inflammatory fat stranding and soft tissue thickening about the abdominal aorta, most notably at the level of the renal arteries. Inflammatory stranding extends along the spinal muscular atrophy (SMA) (Figure 2), consistent with recurrence of aortitis. Steroids were instituted and the aortitis resolved. The patient had subsequent recurrences bud did not respond to treatment with topotecan and subsequent treatment with lutetium Lu 177 dotatate. In December 2020, he had continued disease progression. Treatment with cisplatin, etoposide, and the PD-1 inhibitor pembrolizumab was started. He has had 6 cycles of combined chemoimmunotherapy without recurrence of aortitis and had a partial remission.
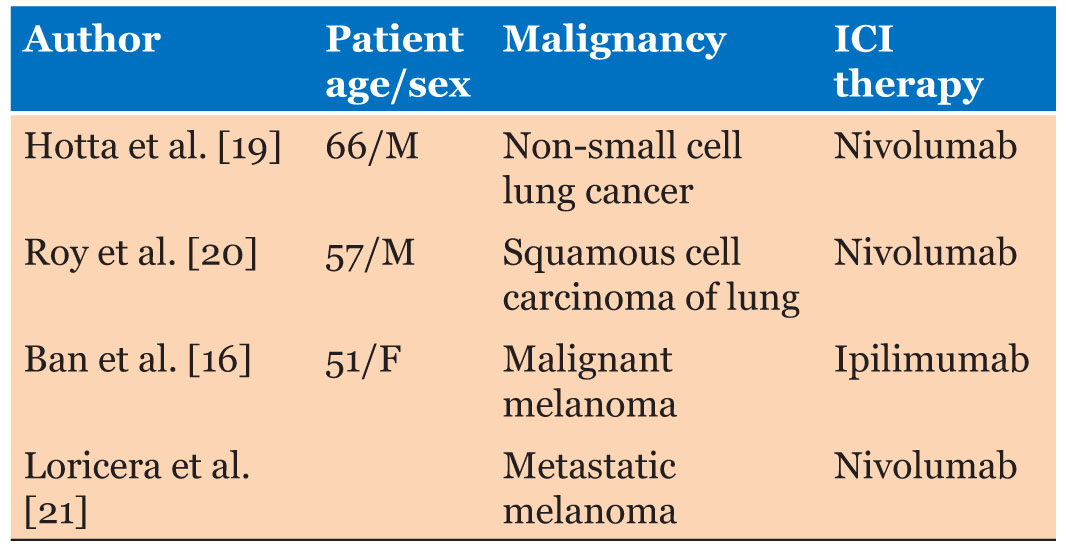
While aortitis is an exceptionally rare reaction when administering immune checkpoint inhibitor therapies, there are four reported cases (Table 1). These cases occurred during treatment with the PD-1 inhibitor nivolumab or the CTLA-4 inhibitor ipilimumab. The patient in this case was treated with atezolizumab, a PD-L1 inhibitor.
Discussion
This patient had a high-grade neuroendocrine tumor of the pancreas with a ki-67 of 90% and RB1 inactivation. He had a much poorer prognosis than low grade neuroendocrine tumors [2],[3],[4],[5]. He was therefore administered with treatment regimens commonly used for small cell lung cancer. The patient originally received 7 cycles of carboplatin and etoposide. After initial response, then rapid recurrence, he was retreated with carboplatin and etoposide but atezolizumab, a selective PD-L1 inhibitor was added. The patient developed aortitis after the second cycle of therapy which promptly resolved with corticosteroids. During third line therapy atezolizumab was again utilized with chemotherapy and again the patient developed aortitis. With 5th line therapy, the PD-1 inhibitor pembrolizumab was utilized with cisplatin and etoposide. The patient received 6 cycles of therapy and had a partial remission without recurrence of aortitis.
The PD-L1 inhibitor atezolizumab was the first immune checkpoint inhibitor FDA approved in small cell cancer and was used first on this patient. We had limited treatment options so when the patient recurred, the PD-1 inhibitor pembrolizumab, which was subsequently approved for recurrent small cell lung cancer, was administered.
While the majority of irAEs are pulmonary, dermatologic, gastrointestinal, hepatic, and endocrine related, large vessel vasculitis, such as aortitis, has been rarely reported with cancer immunotherapy treatments. A PubMed search revealed no prior reports of aortitis following PD-L1 inhibition including the recent 507 patients treated with adjuvant atezolizumab with stage IB-IIIA non-small cell lung cancer and the 201 patients treated with atezolizumab and chemotherapy with small cell lung cancer [6],[7].
Risk factors
This patient had no prior history of collagen vascular disease or other immunologic disease that would increase his risk of immune related adverse events to cancer immunotherapy.
PD-1 and PD-L1 inhibitors
PD-1 is a type I membrane protein, structurally and functionally analogous to the CD-28 receptor family. In various cancers, the PD-1 pathway inhibits the immune response by blocking the expression and action of T-cells.
PD-1 is not expressed on resting T-cells but is induced via phosphorylation by the TCR/MHC and CD28/B7 interactions between T-cells and antigen presenting cells. Once PD-1 is phosphorylated, it inhibits T-cell receptor signals via phosphorylation of Src homology phosphatase-1 (SHP-1) and SHP-22 and hinders T-cell responses to circulating cytokines [8]. Additionally, PD-1 is expressed on regulator T-cells and may be activated to inhibit immune responses by increasing expression of FOXP3, decreasing expression of IFN-γ and producing inhibitory cytokines, such as TGF-β, IL-10 and IL-35 [8]. These effective pro-tumor functions of PD-1 make this receptor a target for immunotherapies in certain malignancies for qualified patients.
PD-1 inhibitors, such as nivolumab and pembrolizumab, target PD-1 receptors on T-cells and prevent the interaction between PD-1 and both of its ligands, PD-L1 and PD-L2. PD-1 inhibition enhances T-lymphocyte function and increases cytokine crosstalk between the PD-1 positive T-cells and dendritic cells specialized in activation in the tumor microenvironment. A consequence of this interaction with PD-L1 and PD-L2 is the release of proinflammatory cytokines, such as TNF-α and IFN-γ, which enhance the stem-like properties of T-cells in the tumor microenvironment [9].
PD-L1 serves as one of the two ligands for PD-1 and is part of the B7 family of type I transmembrane protein receptors. It is commonly expressed by macrophages, activated T-cells and B-cells. PD-L1 is up-regulated in response to T-cell secreted proinflammatory cytokines such as IFN-γ and IL-4 through signal transducer and activator of transcription-1 (STAT1) and IFN regulatory factor-1 (IRF1) [10] in tumor cells for the purpose of evading the immune system response. This upregulation serves as a pro-tumorigenic factor in cancer cells, as PD-L1 binds to its receptor, PD-1, and activates tumor-survival signaling pathways [11].
PD-L1 inhibitors, such as atezolizumab, target the interaction of PD-1 and B7, prevent protumor effects and restore antitumor T-cell function [6]. Additionally, PD-L1 has been shown to exert non-immune proliferative effects on tumor cells [12], providing an additional benefit to anti-PD-L1 therapies. An important difference between PD-1 inhibitors and PD-L1 inhibitors is that PD-L1 inhibitors still allow the interaction between PD-1 and PD-L2. The continued binding of PD-1 to PD-L2 weakens the immune and proinflammatory response and decreases the likelihood of an autoinflammatory adverse reaction. Despite the anti-inflammatory effects of PD-L2 that make these therapies more tolerable, PD-L1 inhibitors, such as atezolizumab, still have significant potential for irAEs, due to their prevention of the PD-1/PD-L1 interaction, and indirect promotion of the release of proinflammatory cytokines. PD-L1 expression is not required in the FDA approval for the use of immune checkpoint inhibitors in small cell lung cancer. The phase III trial of atezolizumab did not require PD-L1 expression for eligibility [7]. Therefore, PD-L1 expression was not assessed in this patient.
This patient’s tumor had a loss of RB1. RB1 may have an important role in modulating expression of genes associated with immune system function [13]. Therefore, RB1 loss may be important in response to immunotherapy. Specific subsets of immune cells were not measured, and his triglycerides and cholesterol were within the institutional normal limits.
Diagnosis
Evaluation of the patient’s history and physical exam, as well as arterial biopsy, computed tomography (CT), magnetic resonance angiography (MRA), Doppler ultrasound, and positron emission tomography (PET) scans have been used to diagnose aortitis [14]. In this case, the patient was diagnosed by evaluation of symptoms when presenting to the emergency department and confirmed via CT scan. Computed tomography, generally using iodinated contrast, is easily available in an emergency setting and can also provide the benefit of ruling out conditions that may mimic aortitis such as aortic dissection, intramural hematoma, and penetrating atherosclerotic ulcer. In the case of acute aortitis, CT may show thickening of the aortic wall and peri-aortic inflammation [15]. Although considered less sensitive for diagnosis, CT is commonly used in follow-up to confirm resolution of the aortitis months after the initial onset of symptoms.
Treatment
Aortitis is commonly treated with high-dose glucocorticoids as well as close monitoring with follow-up imaging, including regular CT scans and blood-pressure monitoring [14]. In rare cases, long-term immunosuppressive therapy may be considered to relieve symptoms and prevent recurrences. When administering glucocorticoids, tapering is utilized to reduce the risk of significant side effects [16]. If the patient does not respond to treatment or if there is a relapse while on corticosteroids, infliximab is recommended [17]. When corticosteroids are used in the management of PD-1/PD-L1 toxicity, the impact on efficacy is uncertain [18].
Conclusion
This is the first case report of a patient developing aortitis following PD-L1 immune blockade. The patient was successfully treated with high dose corticosteroids with each of his two episodes of aortitis with resolution of symptoms within a week. He was successfully treated with six cycles of a PD-1 inhibitor pembrolizumab without recurrence of aortitis.
REFERENCES
1.
Alsaab HO, Sau S, Alzhrani R, et al. PD-1 and PD-L1 checkpoint signaling inhibition for cancer immunotherapy: Mechanism, combinations, and clinical outcome. Front Pharmacol 2017;8:561. [CrossRef]
[Pubmed]

2.
Mafficini A, Scarpa A. Genetics and epigenetics of gastroenteropancreatic neuroendocrine neoplasms. Endocr Rev 2019;40(2):506–36. [CrossRef]
[Pubmed]

3.
Maru DM, Khurana H, Rashid A, et al. Retrospective study of clinicopathologic features and prognosis of high-grade neuroendocrine carcinoma of the esophagus. Am J Surg Pathol 2008;32(9):1404–11. [CrossRef]
[Pubmed]

4.
Ishida M, Sekine S, Fukagawa T, et al. Neuroendocrine carcinoma of the stomach: Morphologic and immunohistochemical characteristics and prognosis. Am J Surg Pathol 2013;37(7):949–59. [CrossRef]
[Pubmed]

5.
Ikeda M, Okuyama H, Takahashi H, et al. Chemotherapy for advanced poorly differentiated pancreatic neuroendocrine carcinoma. J Hepatobiliary Pancreat Sci 2015;22(8):623–7. [CrossRef]
[Pubmed]

6.
Wakelee HA, Altorki NK, Zhou C, et al. IMpower010: Primary results of a phase III global study of atezolizumab versus best supportive care after adjuvant chemotherapy in resected stage IB-IIIA nonsmall cell lung cancer (NSCLC). Journal of Clinical Oncology 2021;39(15_Suppl):8500. [CrossRef]

7.
Horn L, Mansfield AS, Szczęsna A, et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N Engl J Med 2018;379(23):2220–9. [CrossRef]
[Pubmed]

8.
Akinleye A, Rasool Z. Immune checkpoint inhibitors of PD-L1 as cancer therapeutics. J Hematol Oncol 2019;12(1):92. [CrossRef]
[Pubmed]

9.
Berraondo P. Mechanisms of action for different checkpoint inhibitors. HemaSphere 2019;3(S2):28–30. [CrossRef]

10.
Chen J, Jiang CC, Jin L, Zhang XD. Regulation of PDL1: A novel role of pro-survival signalling in cancer. Ann Oncol 2016;27(3):409–16. [CrossRef]
[Pubmed]

11.
Dong P, Xiong Y, Yue J, Hanley SJB, Watari H. Tumor-intrinsic PD-L1 signaling in cancer initiation, development and treatment: Beyond immune evasion. Front Oncol 2018;8:386. [CrossRef]
[Pubmed]

12.
Han Y, Liu D, Li L. PD-1/PD-L1 pathway: Current researches in cancer. Am J Cancer Res 2020;10(3):727–42.
[Pubmed]

13.
Knudsen ES, Pruitt SC, Hershberger PA, Witkiewicz AK, Goodrich DW. Cell cycle and beyond: Exploiting new RB1 controlled mechanisms for cancer therapy. Trends Cancer 2019;5(5):308–24. [CrossRef]
[Pubmed]

14.
Gornik HL, Creager MA. Aortitis. Circulation 2008;117(23):3039–51. [CrossRef]
[Pubmed]

15.
Kissin EY, Merkel PA. Diagnostic imaging in Takayasu arteritis. Curr Opin Rheumatol 2004;16(1):31–7. [CrossRef]
[Pubmed]

16.
17.
Friedman CF, Proverbs-Singh TA, Postow MA. Treatment of the Immune-related adverse effects of immune checkpoint inhibitors: A review. JAMA Oncol 2016;2(10):1346–53. [CrossRef]
[Pubmed]

18.
Brahmer JR, Lacchetti C, Schneider BJ, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol 2018;36(17):1714–68. [CrossRef]
[Pubmed]

19.
Hotta M, Naka G, Minamimoto R, Takeda Y, Hojo M. Nivolumab-induced periaortitis demonstrated by FDG PET/CT. Clin Nucl Med 2020;45(11):910–2. [CrossRef]
[Pubmed]

20.
Roy AK, Tathireddy HR, Roy M. Aftermath of induced inflammation: Acute periaortitis due to nivolumab therapy. BMJ Case Rep 2017;2017:bcr2017221852. [CrossRef]
[Pubmed]

21.
Loricera J, Hernández JL, García-Castaño A, Martínez-Rodríguez I, González-Gay MÁ, Blanco R. Subclinical aortitis after starting nivolumab in a patient with metastatic melanoma. A case of drug-associated aortitis? Clin Exp Rheumatol 2018;36 Suppl 111(2):171.
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Nicholas R Liguori - Conception of the work, Design of the work, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Michael V Longo - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Howard Safran - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestThe authors declare that the case report production was conducted in the absence of any commercial or financial relationships that would classify as a conflict of interest.
Copyright© 2021 Nicholas R Liguori et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.