|
Case Report
Rare case of recurrent chylothorax following irradiation for esophageal cancer
1 Department of Medical Oncology, University Hospital Limerick, Limerick, Ireland
2 Mid-Western Radiation Oncology Centre, Limerick, Ireland
3 Department of Respiratory Medicine, University Hospital Limerick, Ireland
4 Department of Thoracic Surgery, Mater Misericordiae University Hospital, Dublin, Ireland
Address correspondence to:
Ruba Ahmed Hamed
Midwest Cancer Center, Oncology Department, University Hospital Limerick,
Ireland
Message to Corresponding Author
Article ID: 100086Z10RH2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Hamed RA, Mahgoub T, Elbassiouni M, O’Brien A, Redmond KC, Korpanty G. Rare case of recurrent chylothorax following irradiation for esophageal cancer. J Case Rep Images Oncology 2021;7:100086Z10RH2021.ABSTRACT
Introduction: Chylothorax is a rare cause of pleural effusion that results from damage of the thoracic duct and chyle leakage from the lymphatic system into the pleural space. Causes of chylothorax can be traumatic or non-traumatic. Chylothorax occurring due to damage to the thoracic duct by thoracic irradiation is rare, and infrequently described in the literature.
Case Report: We present a case of a 74-year-old patient with a history of stage IV esophageal cancer who developed recurrent bilateral chylothoraces after palliative radiotherapy to her esophagus. She was treated conservatively with multiple modalities, including total parental nutrition leading to an oral low-fat medium-chain triglyceride diet, subcutaneous octreotide injection, and long-term insertions of bilateral chest drains.
Conclusion: This case report and discussion highlight that chylothorax is a rare complication of radiotherapy. A suspicion of chylothorax should be raised in patients who present with recurrent pleural effusions after radiotherapy to the esophagus or mediastinum, as early intervention can improve outcomes in these patients.
Keywords: Chylothorax, Esophageal cancer, Radiotherapy
Introduction
Chylothorax is an accumulation of chyle (lymphatic fluid) in pleural space that occurs secondary to damage of the thoracic duct system with chyle leakage. It contains a high concentration of triglycerides in the form of chylomicrons, as well as lymphocytes and immunoglobulins [1]. The etiology of chylothorax can be classified as traumatic or non-traumatic. Traumatic cases can be iatrogenic or non-iatrogenic (20% of traumatic cases) [1].
Thoracic surgery is the leading cause of trauma with esophageal surgery reporting an incidence of 4% [2]. Other iatrogenic traumatic causes include thoracic duct damage following subclavian vein catheterization and duct blockage due to venous thrombosis secondary to central venous catheterization [1],[2].
Non-iatrogenic traumatic causes include thoracic duct damage following fracture dislocation of the spine, thoracic duct injury following childbirth, and penetrating trauma.
Non-traumatic etiologies include malignancy, sarcoidosis, amyloidosis, superior vena cava thrombosis, benign tumors, retrosternal goiter, congenital duct abnormalities, and diseases of the lymph vessels such as yellow nail syndrome, hemagiomatosis, and lymphangioleiomyomatosis [1].
Thoracic duct obstruction due to malignancy is the commonest cause of non-traumatic chylothorax with lymphoma accounting for about 70% of the cases. Chylothorax has also been described as an early and late complication of radiotherapy due to damage to the thoracic duct by radiation [1],[2].
Diagnostic criteria of chylothorax involve presence of chylomicrons, cholesterol concentration <5.18 mmol/L, and triglyceride >1.24 mmol/L in the pleural fluid [1].
Treatment of chylothorax may be either conservative or surgical depending on the clinical scenario [1].
Conservative treatment includes oral dietary fat-restriction foods with supplement of medium-chain triglycerides or fasting with central venous nutrition, pharmacotherapy by administering octreotide, synthetic somatostatin analogue, or etilefrine which stimulates the sympathetic nervous system to inhibit production of chyle and decrease lymph flow in the thoracic duct, or performing a chemical pleurodesis such as tetracycline or minomycin [3].
Surgical treatment involves thoracic duct ligation by video-assisted thoracic surgery (VATS), or percutaneous thoracic duct cannulation and insertion of an indwelling metal coil for thoracic duct embolization [3],[4].
We report an uncommon case of recurrent chylothorax which occurred after palliative radiotherapy to the esophagus in a patient with stage IV esophageal cancer. The cause of chylothorax in our case was believed to be due to the damage of the thoracic duct by radiotherapy delivered six months earlier. The patient was treated conservatively with medium-chain triglyceride low-fat diet, octreotide injections, and pleurocentesis allowing an auto-pleurodesis.
Case Report
A 74-year-old lady was diagnosed with stage IV moderately differentiated squamous cell carcinoma of the mid and lower esophagus with metastatic disease in the right supraclavicular and retroperitoneal lymph nodes in August 2018.
She was treated with palliative radiotherapy to the mid esophagus with 40 Gy/10 fractions in September 2018 followed by four cycles of palliative chemotherapy with carboplatin and paclitaxel.
A restaging computed tomography (CT) scan after completion of chemotherapy demonstrated resolution of her lymphadenopathy; however, she developed new bilateral pleural effusions. The scan also demonstrated new mild to moderated pericardial effusion which was re-assessed by echocardiogram that confirmed small pericardial effusion with normal systolic and diastolic functions.
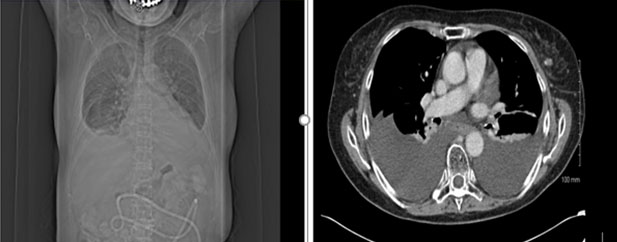
She developed exertional dyspnea and underwent bilateral pleurocentesis with 700 mL fluid drained from her right side and 1500 mL from her left side. The fluid appeared yellow and thick and cytology of both showed no malignant cells (Figure 1).
A month later she was admitted with dyspnea, right sided chest pain, and hypoxia. Her vital signs showed low oxygen saturation at 88–90% in room air, blood pressure and pulse rate were within acceptable ranges. She required 2–4 liters of oxygen/min to maintain saturation of 94%. A chest radiograph showed recurrence of bilateral pleural effusions, larger on the right side (Figure 2).
She had a right chest drain interested. Analysis of the pleural fluid showed exudative picture with pH 7.7, high protein 50 g/L, albumin 24 g/L, glucose 6.4 mmol/L, amylase 25 IU/L, lactate dehydrogenase (LDH) 110 U/L, high leukocytes count 310/uL mainly mononuclear at 95%. The suspicion of chylothorax was raised and further analysis of pleural fluid revealed cholesterol level at 2 mmol/L and triglycerides 4 mmol/L.
The diagnosis of chylothorax was confirmed on the basis of the fluid biochemistry, with cholesterol 1.24 mmol/L diagnostic of chylous pleural fluid. Fluid chylomicron concentration was not available.
The patient was managed initially conservatively with medium-chain triglyceride low-fat diet and total parenteral nutrition. She was also commenced on octreotide, initially at 50 mcg three times a day (TDS) subcutaneously and then increased to 100 mcg TDS. She also had 4 liters of pleural fluid drained from her right lung. A chest radiograph after completion of 14 days of octreotide injections showed an increasing right-sided pleural effusion. Computed tomography of the thorax also confirmed progression of a pleural effusion, as well as her cancer (Figure 3).
She was then referred to the thoracic surgery service. She was not deemed suitable for invasive interventional approaches due to her progressive disease, and she underwent bilateral long-term tunneled indwelling drain insertions with PleurX catheters. She was transferred to a hospice with drains remained in situ, and where she passed away two months later.
Discussion
Malignancy-associated chylothorax is rare; however, it is the commonest cause of non-traumatic chylothorax, with lymphoma accounting for 70% of cases among the neoplastic etiologies for chylothorax [1]. Esophageal resection is the most common iatrogenic cause of chylothorax, with an incidence of 4% reported in the literature [2]. Chylothorax can also occur as both an early or late complication of mediastinal radiotherapy when administered for a multitude of indications [2]. It has been reported following radiotherapy for Hodgkin’s lymphoma, soft-tissue sarcoma, non-small-cell lung cancer, esophageal cancer, and otherwise mediastinal metastatic diseases. The mechanism of thoracic duct obstruction/disruption post?irradiation is uncertain but may relate to lymphatic obstruction due to mediastinal fibrosis [5].
Post-radiotherapy chylothorax has been described infrequently in the medical literature and there have been few case reports following irradiation to the neck and/or thorax [5],[6],[7],[8]. In our case, chylothorax was confirmed based on pleural fluid analysis, and fulfillment of the diagnostic criteria. We believe that damage of the thoracic duct from radiotherapy was the main mechanism of our patient’s recurrent pleural effusions.
A previous study published in 1997 [6] described a case of a 46-year-old lady who developed chylous ascites and bilateral chylothorax secondary to previous radiotherapy to her chest and abdomen for Hodgkin’s lymphoma.
A more recent case report [7] published in 2003 reported a case of a 64-year-old lady with stage IV middle esophageal squamous cell carcinoma who developed chylothorax two years after esophageal irradiation and was treated with VATS and thoracic-duct clipping. This is similar to our case; however, our patient was managed differently with a more palliative conservative approach due to her progressive disease and poor performance status. PleurX catheters exclude the need for a general anesthetic and allows for discharge planning from an acute hospital setting either to the hospice or home while achieving an effective auto-pleurodesis.
Conclusion
Overall, chylothorax is a rare complication that can result from radiotherapy. This diagnosis should be considered in the differential diagnosis in patients present with recurrent pleural effusion after radiotherapy to the esophagus or mediastinum. To our knowledge there is only one other case report which describes radiotherapy related chylothorax in esophageal cancer.
REFERENCES
1.
McGrath EE, Blades Z, Anderson PB. Chylothorax: Aetiology, diagnosis and therapeutic options. Respir Med 2010;104(1):1–8. [CrossRef]
[Pubmed]

2.
Nair SK, Petko M, Hayward MP. Aetiology and management of chylothorax in adults. Eur J Cardiothorac Surg 2007;32(2):362–9. [CrossRef]
[Pubmed]

3.
Hayashibara N, Ogawa T, Tsuji E, Ishizuna K. Efficacy of octreotide against chylothorax following lateral neck dissection for thyroid cancer: A case report. Int J Surg Case Rep 2016;21:107–10. [CrossRef]
[Pubmed]

4.
5.
McWilliams A, Gabbay E. Chylothorax occurring 23 years post-irradiation: Literature review and management strategies. Respirology 2000;5(3):301–3. [CrossRef]
[Pubmed]

6.
Promisloff RA, Hogue DJ. Chylothorax: The result of previous radiation therapy? J Am Osteopath Assoc 1997;97(3):164–6. [CrossRef]
[Pubmed]

7.
Thomson AH, Sivalingham S, Rajesh PB, Elyan SAG. Chylothorax after radiotherapy in oesophageal carcinoma. Lancet Oncol 2003;4(11):703–4. [CrossRef]
[Pubmed]

8.
Van Renterghem D, Hamers J, De Schryver A, Pauwels R, Van der Straeten M. Chylothorax after mantle field irradiation for Hodgkin’s disease. Respiration 1985;48(2):188–9. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Ruba Ahmed Hamed - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Thamir Mahgoub - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mazen Elbassiouni - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Aidan O'Brien - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Karen C Redmond - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Greg Korpanty - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Ruba Ahmed Hamed et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.