|
|
Case Report
| ||||||
| Primary melanoma of the vagina: A review of treatment strategies | ||||||
| Moulton L.1, Goodrich S.2, Rose P.G.2 | ||||||
|
1Ob/Gyn and Women's Health Institute, Cleveland Clinic, Cleveland, OH, USA.
2Gynecologic Oncology division, Ob/Gyn and Women's Health Institute, Cleveland Clinic, Cleveland, OH, USA. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article: |
| Moulton L, Goodrich S, Rose PG. Primary melanoma of the vagina: A review of treatment strategies. J Case Rep Images Oncology 2017;3:1–5. |
|
Abstract
|
|
Introduction:
Melanomas of the female lower genital tract are rare, but account for approximately 10% of melanomas diagnosed in women. The five-year survival rate for vaginal melanoma is low, approximating 18%. Due to the rarity of this disease and poor prognosis, there is limited information for evidence-based treatment strategies.
Case Report: This case report discusses the management of a patient diagnosed with primary vaginal melanoma treated with total pelvic exenteration with negative margins, followed by adjuvant chemotherapy with temozolomide and cisplatin. Six months following the completion of chemotherapy, the patient developed liver metastasis. Conclusion: While adjuvant chemotherapy with temozolomide and cisplatin showed promise due to improved recurrence free survival compared to observation and high-dose interferon in patients with mucosal melanomas, further studies are needed to determine an effective adjuvant therapy regimen in patients with vaginal melanoma. | |
|
Keywords:
Adjuvant, Chemotherapy, Exenteration, Melanoma, Pelvic, Vaginal
| |
|
Introduction
| ||||||
|
Malignant melanoma impacts a significant number of lives in the United States with 76,100 new diagnoses in 2014 [1] [2]. Among the subtypes of melanomas, cutaneous melanomas are the most common and have the highest overall survival rate, estimated at 98.1% at five years for localized disease. Mucosal melanomas are less common, comprising about 1% of all melanomas and they generally have a worse prognosis. Within the female lower genital tract, 95% of melanomas arise in the vulva, and 3% originate within the vagina [1]. Primary vaginal melanomas are associated with worse outcomes compared to those that originate in the vulva. The estimated five-year survival for primary vaginal melanoma is 18% compared to 47% for vulvar melanoma [3]. There is limited data for evidence-based management of vaginal melanoma due to the low prevalence and dismal overall survival. We present a case report of a patient with primary vaginal melanoma treated with total pelvic exenteration with negative margins, followed by adjuvant chemotherapy with temozolomide and cisplatin. | ||||||
|
Case Report
| ||||||
|
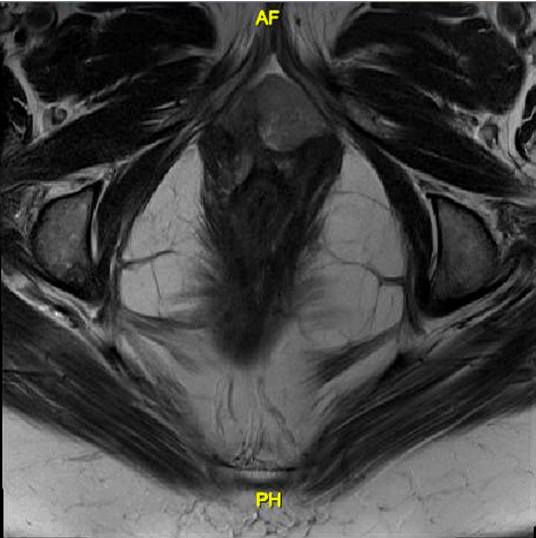
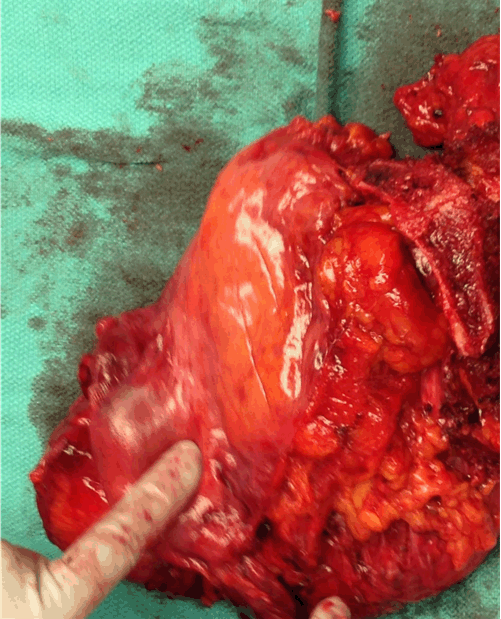
A 57-year-old female presented with complaint of abnormal vaginal drainage and was found to have a vaginal mass measuring about 3–4 cm. Her past medical history was significant for stage III colon cancer treated with surgical resection and adjuvant chemotherapy with no evidence of recurrence. She denied family history of skin cancer or gynecologic malignancy. Social history was significant for 10 years of tobacco use. Biopsy of the lesion showed a high-grade malignant neoplasm consistent with melanoma. Immunohistochemical analysis of this specimen was positive for S-100, HMB45, MART-1, weakly positive for CAM 5.2 and negative for P16, P63, CK-7, CK-20. The patient was referred to gynecologic oncology for further evaluation. Pelvic examination demonstrated a 4-cm mass replacing the anterior distal vaginal mucosa and urethra. On pelvic MRI scan, a lobulated soft tissue mass measuring 2.4x1.8x2.9 cm extending from the posterior wall of the left vaginal fornix and prolapsing inferiorly into the vaginal canal was present. The mass appeared to infiltrate into the left vaginal forniceal wall superiorly (Figure 1). Whole body PET scan demonstrated an intensely hypermetabolic left anterolateral vaginal lesion measuring 2.8x2.1 cm consistent with a neoplastic process with no inguinal or pelvic lymphadenopathy. Since these images showed no evidence of metastatic disease, malignancy was presumed to be of primary vaginal origin. The patient underwent a bilateral inguinofemoral lymphadenectomy and bilateral pelvic lymphadenectomy with negative nodes followed immediately by a total pelvic exenteration to ensure negative surgical margins with resection of pubic bone, colostomy and diverting ileal urinary conduit. Intraoperative findings were significant for 4–5 cm exophytic anterior vaginal mass located 1 cm left of the urethra (Figure 2). Frozen section of the resected bilateral inguinofemoral and pelvic lymph nodes were negative for malignancy. Final pathology demonstrated a 3.5cm melanoma with ulceration invading into the posterior wall of the vagina to a maximum depth of 20 mm. Based on these findings, the patient had AJCC stage IIC disease. There was no evidence of metastasis to the inguinal or pelvic lymph nodes bilaterally. The bladder, anus, rectum, urethra, cervix, uterus, ovaries were negative for malignancy. Following a consultation with medical oncology, she was started on adjuvant chemotherapy with temozolomide 200 mg/m2/d. Days 1–5 plus cisplatin 75 mg/m2 IV divided into three days, repeated every three weeks for six cycles. After cycle #5, she received 20% dose reduction of cisplatin due to thrombocytopenia. After completion of six cycles, PET/CT demonstrated no metastatic disease. Six months following the completion of chemotherapy, hypermetabolic activity was noted within the liver on a surveillance whole body PET scan, and subsequently confirmed to be metastatic melanoma with liver biopsy. After metastatic disease was identified, the patient was started on immunotherapy with ipilimumab and nivolumab. After two cycles, she developed a grade 3 macular erythematous rash necessitating corticosteroid therapy and cessation of ipilimumab. After three additional cycles of nivolimuab, the patient has no further toxicity and no evidence of disease progression. | ||||||
|
| ||||||
| ||||||
|
Discussion
| ||||||
|
There is limited data for evidence-based management of vaginal melanoma due to the rarity of the disease and the poor overall survival. Many of the studies on this topic are retrospective, have small sample sizes or combine data for all mucosal melanomas or both vulvar and vaginal melanomas, which have significantly different overall survival rates [3]. Surgical excision is important in the initial management of vaginal melanoma, but controversy exists regarding whether radical or conservative surgery leads to better outcomes. Several studies have shown that initial radical surgery may benefit patients without metastatic disease. Chung et al. achieved local control in five of seven women who had radical surgery, and three were alive without disease at 5.5 to 16 years. In this same study, three patients who had wide local excision developed disease recurrence at 19, 26 and 69 months after surgery [4]. In a study of eight patients by Van Nostrand et al., there was a significant improvement in survival at two years among the patients who underwent radical surgery (48%) compared to those who had conservative surgery (20%) [5]. Similarly, in a case series by Geisler et al., four patients with invasive vaginal melanoma and no metastatic disease were treated with pelvic exenteration and had no recurrence after 31 to 97 months [6]. However, recent studies support conservative surgical practices for resection of vaginal melanoma. Frumovitz et al., demonstrated a small, not statistically significant difference in overall survival of five months between patients who had pelvic exenteration compared to those who had conservative surgery with wide local excision [7]. In a retrospective series at Mayo Clinic, radical surgery was not associated with improved overall survival compared to conservative surgery. This conflicting data demonstrates the need for further research evaluating the optimal surgical strategy in patients with vaginal melanoma [8]. The decision for radical versus conservative surgery should be made after determination that the patient has no metastatic disease. A thorough discussion of the associated morbidities of radical exenterative surgery should occur between the patient and physician. There are no established guidelines for neoadjuvant or adjuvant treatment of vaginal melanoma. Therefore, current research into neoadjuvant and adjuvant therapies for vaginal and mucosal melanomas is largely derived from cutaneous melanoma studies. In cutaneous melanomas, patients who are at high-risk for recurrence, generally, defined as AJCC IIB-III stage disease, are considered for adjuvant therapy, which may consist of any combination of systemic chemotherapy, immunotherapy or biochemotherapy [9]. Several randomized trials have identified that adjuvant immunotherapy with interferon-alpha improves overall survival and relapse-free survival in high-risk patients with cutaneous melanomas and this has been a mainstay of therapy for many years [10] [11]. Systemic chemotherapy has also been explored for adjuvant treatment of cutaneous melanomas. Dacarbazine is considered the most active single chemotherapy agent for metastatic melanoma with response rates between 8–20% [12]. Temozolomide is the oral analog of dacarbazine and when given with or without cisplatin, it has equal efficacy and possibly improved quality of life compared to dacarbazine [13]. Several of these regimens have been used in patients with vaginal melanoma with variable response. In a recent retrospective review from Mayo Clinic, one patient treated with two cycles of temozolomide had no clinical benefit, but another treated with neoadjuvant carboplatin/paclitaxel followed by radical surgery and bevacizumab was disease free at fifth year [8]. Harting et al. identified 11 patients treated with biochemotherapy for advanced disease, consisting of a combination of cisplatin, vinblastine, dacarbazine, high-dose interferon alpha and interleukin-2 with an overall response rate of 36% and median overall survival of 10 months [14]. Frumovitz et al. reported a possible role of radiation for neoadjuvant or adjuvant therapy of women with primary vaginal melanoma. Of 20 patients who had wide local excision, patients who received adjuvant radiation had a median survival of 29.4 months, compared to 16.1 months in those who did not [7]. The only data that demonstrates efficacy of systemic chemotherapy in mucosal melanomas specifically is from a phase II clinical trial in China. This trial randomized patients with adequately resected mucosal melanomas to high dose interferon alfa-2b or temozolomide with cisplatin for systemic adjuvant therapy. Of 189 patients with Stage II/III mucosal melanoma without evidence of distant metastasis, median recurrence free survival was 5.4 months in the observation group, compared to 9.4 months in the high-dose interferon group and 20.8 months in the temozolomide plus cisplatin group. Similarly, temozolomide plus cisplatin significantly improved overall survival compared to high dose interferon without significantly increased toxicity [15]. | ||||||
|
Conclusion
| ||||||
|
To our knowledge, this is the first report of a patient with vaginal melanoma treated with radical surgery, followed by adjuvant chemotherapy with this regimen. In this case, the recurrence occurred at sixth month after completion of treatment. While adjuvant chemotherapy with temozolomide and cisplatin showed promise due to improved recurrence free survival compared to observation and high-dose interferon in patients with mucosal melanomas, this regimen needs further study in patients with vaginal melanoma specifically. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Moulton L. – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Goodrich S. – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Rose P.G. – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Moulton L. et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|