| Table of Contents |  |
|
Clinical Image
| ||||||
| Cachexia, vomiting and an unexpected abdominal mass | ||||||
| João Pedro Tavares1, Suzana Natividade Calretas1, Rui Marques dos Santos2, Armando Carvalho2 | ||||||
|
1MD, Internal Medicine Department, Centro Hospitalar e Universitário de Coimbra, Coimbra, Portugal.
2MD PhD, Internal Medicine Department, Centro Hospitalar e Universitário de Coimbra, Coimbra, Portugal. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Tavares JP, Calretas SN, dos Santos RM, Carvalho A. Cachexia, vomiting and an unexpected abdominal mass. J Case Rep Images Oncology 2016;2:32–34. |
|
Case Report
| ||||||
|
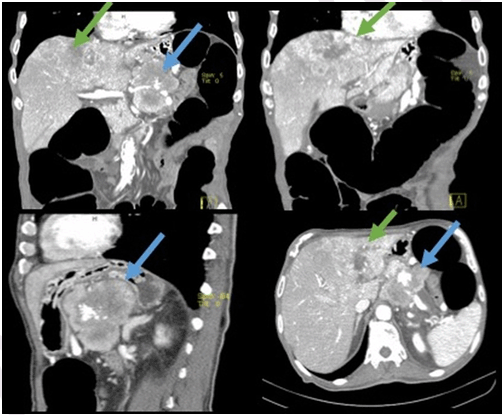
An 86-years-old male was admitted with a weight loss of 28% in the last 10 months, with a two-month history of anorexia and recurrent postprandial vomiting (2–3/day). Four months earlier, he sought medical attention complaining of unspecific abdominal symptoms. The patient's blood tests, abdominal X-ray and colonoscopy, were all normal. He had a past medical history of type 2 diabetes mellitus, hypertension and dyslipidemia, under treatment with insulin, candesartan and simvastatin. Physical examination was remarkable for cachexia, with a body mass index of 15.7 kg/m2 and a solid, irregular and painless mass in the epigastrium. Blood tests were positive for a normocytic normochromic anemia, γGT 1.5x upper limit of normal and HbA1c of 8%. Chromogranin A value of 1137 ng/mL (reference range <85). Abdomen ultrasound revealed a lobulated heterogeneous mass in the epigastrium with a hypoechogenic nodule extending to both the liver lobes. An abdominal computed tomography scan (Figure 1) revealed 8x6 cm mass arising from the tail of the pancreas, with internal calcifications, which entraped and compressed splenic vessels. Computed tomography scan showed an heterogenous liver mass, resulting of confluent nodules extending to both hepatic lobes in a pattern compatible with a heavy burden of hepatic metastatic disease. 68Gallium-DOTA-NOC PET-scan confirmed computed tomography scan findings and revealed extensive ganglionic, lung, bone and peritoneal involvement. Emphasizing the pancreatic mass as having a highly proliferative behavior since it had a low expression of somatostatin receptors and also revealing a divergent metastatic behavior witch had a high expression of these receptors. A liver nodule biopsy confirmed the diagnosis of a pancreatic neuroendocrine tumor (pNET), and graded it as a G2 tumor. During the admission period, vomiting improved with metoclopramide. The patient died three weeks after admission as a result of urosepsis. | ||||||
| ||||||
|
Discussion
| ||||||
|
The pNETs are a rare type of pancreatic neoplasms, with an incidence of 0.22–0.32/100000/year. It has a slight male predominance and are more frequent in individuals aged over 55 years [1] [2] [3]. They can be classified as functioning (40–55%) or non-functioning (45–60%) [1][2]. Most pNETs are sporadic and symptoms can arise either from the local mass effect and/or secreted hormones. Diagnosis is usually late with 50% of patients presenting with liver metastasis, and relies on an exhaustive search/discretion of the presenting symptoms, physical examination, laboratory evaluation with relevant biomarkers (chromogranin A, neuron-specific enolase, pancreatic polypeptide and specific hormones related to the clinical presentation: gastrin for patients with Zollinger-Ellison's syndrome, insulin for hypoglycemic syndromes, glucagon for glucagonoma syndrome and VIP for the Verner-Morrison syndrome); and imaging studies, namely CT scan, magnetic resonance imaging scan, and somatostatin receptor scintigraphy or more recently 68Gallium-DOTA-TOC/-NOC/-TATE PET scan. Histology is required for diagnostic confirmation and tumor grading [1] [3]. Surgery is the mainstay of curative treatment [1] [3]. Procedures such as transarterial embolization and radiofrequency ablation are often employed to treat unresectable liver metastasis [1]. Pharmacologic treatment relies on somatostatin analogs and chemotherapy [3]. Prognosis is based on clinical stage, with a five-year survival in well-differentiated tumors of 29% and 41% for non-functioning and functioning neoplasms, respectively [2]. The presented case highlights the diagnostic challenge of pNETs, since the patient presented with unspecific abdominal complaints and was previously evaluated without reaching a diagnosis, two months before the development of compressive symptoms (vomiting). | ||||||
|
Conclusion
| ||||||
|
Pancreatic neuroendocrine neoplasms are a rare type of cancer with a highly variable presentation, ranging from local symptoms to hormone mediated paraneoplastic syndromes. A high index of suspicion is required as well as a careful history taken. Blood tests, imaging and histology studies confirm the diagnosis. Curative resection of the primary tumor and locoregional lymph node metastases improves outcomes in these patients. Keywords: Abdominal mass, Anorexia, Cachexia, Liver metastasis, Pancreatic neoplasia, Pancreatic neuroendocrine tumor, Vomiting | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
João Pedro Tavares – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Suzana Natividade Calretas – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Rui Marques dos Santos – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Armando Carvalho – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2016 João Pedro Tavares et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|