| Table of Contents |  |
|
Case Report
| ||||||
| Recurrent triple negative breast cancer with early severe systemic metastases | ||||||
| Sabrina S. Sam1, Karel T.S. Valenta1 | ||||||
|
1MS3, Northwest Hospital, Randallstown, MD, USA.
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article: |
| Sam SS, Valenta KTS. Recurrent triple negative breast cancer with early severe systemic metastases. J Case Rep Images Oncology 2016;2:14–17. |
|
Abstract
|
|
Introduction:
Triple negative breast cancer is an aggressive subtype seen in up to 20% of breast cancer diagnoses. The locations of metastases differ from receptor positive breast cancers and recurrence occurs earlier after treatment.
Case Report: Our patient is a 65-year-old female diagnosed with stage II high grade triple negative invasive ductal carcinoma after a suspicious abnormality was found on a screening mammogram. She received neoadjuvant chemotherapy, bilateral total mastectomy, and radiation therapy. Two months after radiation was completed she developed nodules over her chest wall and was subsequently diagnosed with recurrent breast cancer with metastases to the skin, lungs, and bone. Conclusion: Triple negative breast cancer metastasizes most commonly to the brain and lungs. Distant spread is also seen more frequently and earlier. Altered follow-up protocols for patients with triple negative breast cancer are essential in order to identify metastases earlier. | |
|
Keywords:
Metastatic breast cancer, Sternal metastasis, Triple negative breast cancer
| |
|
Introduction
| ||||||
|
Triple negative breast cancer (TNBC) refers to tumors that do not express progesterone or estrogen receptors (PR/ER) or human epidermal growth factor receptor factor 2 (HER2). This subtype occurs in 15–20% of breast tumors. The TNBC metastases often occur without prior local recurrence and there is a higher prevalence of brain metastases [1]. According to a study at Women's College Hospital in Toronto, patients with TNBC were more likely to have high grade (66% versus 28%) and larger tumors (3.0 cm versus 2.1 cm) with more lymph node involvement (54.6% versus 45.6%) at diagnosis. These patients had a higher mortality (42.2% versus 28%) and all deaths occurred within 10 years of diagnosis. There were more distant recurrences (33.9% versus 20.4%) occurring sooner in patients with TNBC (2.6 versus 5.0 years) [2]. | ||||||
|
Case Report
| ||||||
|
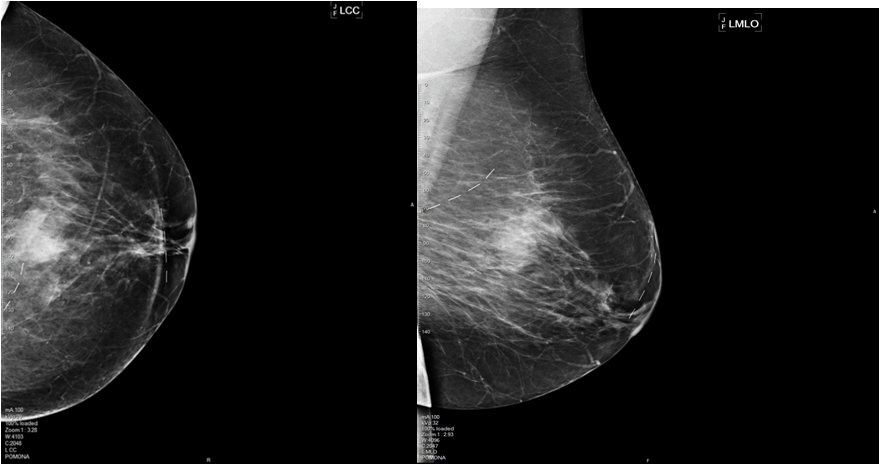
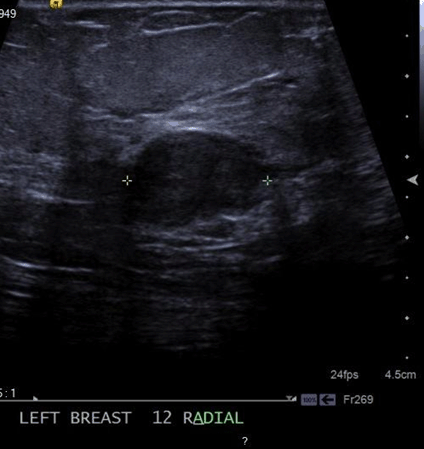
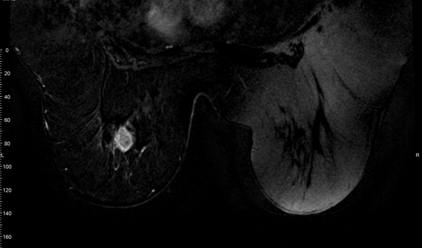
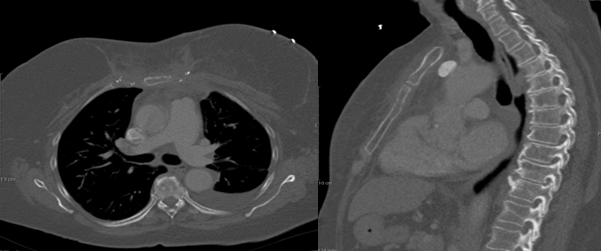
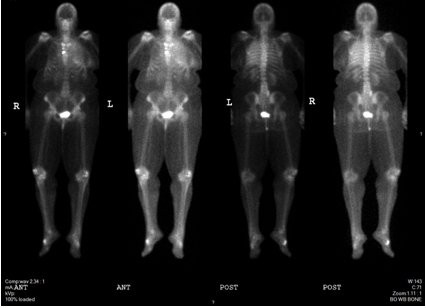
A 65-year-old female was presented with an abnormal screening mammogram (Figure 1). An ultrasound guided core needle biopsy was positive for invasive ductal carcinoma (IDC) with a grade of 3 on the Nottingham/Elston scale (Figure 2). The specimen was triple negative and had a Ki-67 of 90%. Magnetic resonance imaging (MRI) confirmed a 2.5 cm solid mass (Figure 3). A sentinel lymph node biopsy with radiolabeled colloid and isosulfan blue was performed two months after the screening mammogram and was negative for carcinoma completing staging as triple negative IIA (T2, N0) IDC. Neoadjuvant chemotherapy was chosen to begin treatment due to the size, high grade, triple negative status, and in order to directly observe the effect on the tumor. Six treatments of cyclophosphamide and docetaxel resulted in reduction of tumor size to 1.4 cm. Our patient was a candidate for breast conservation but was adamant that she receive a total mastectomy. Six weeks later she had a bilateral total mastectomy with transverse rectus abdominis muscle (TRAM) reconstruction. The surgical specimen displayed poorly differentiated IDC with a residual tumor of 1.4 cm and clear margins of at least 5 mm. Lymphovascular invasion was present. Four months after the procedure, she underwent radiation therapy to the left chest wall with 45 Gy in 25 fractions. This was deemed necessary due to the lymphovascular space involvement. Three months after radiation was completed, the patient presented to the emergency department (ED) with a one month history of increasing chest wall nodules and constant severe pain across the upper left chest. The pain was exacerbated by movement and inspiration and she was tender in her presternal area without evidence of lymphadenopathy by palpation. Computed tomography (CT) scan of the thorax confirmed destruction of the sternum and manubrium with soft tissue swelling anteriorly (Figure 4). Pulmonary nodules and lymphadenopathy measuring 1.6 mm in the retrocaval, 2.4 mm in the subcarinal and 2 mm in the hilar lymph nodes were evident. A punch biopsy of the chest wall nodules confirmed poorly differentiated IDC consistent with her primary breast cancer. Her bone scan confirmed that bony metastases were limited to the sternum and manubrium (Figure 5). She then began systemic therapy with gemcitabine and carboplatin. Her systemic disease with triple negative status and symptomatic visceral crisis directed the need for chemotherapy as per national comprehensive cancer network (NCCN) guidelines. Our patient has not yet received imaging of the head. | ||||||
|
| ||||||
| ||||||
| ||||||
|
| ||||||
| ||||||
|
Discussion
| ||||||
|
Our patient had an uncommon appearance for recurrent breast cancer with early and severe systemic metastases. Her recurrence occurred only two months after radiation therapy was completed and presented with metastases to the lungs, skin and bone. Receptor positive breast cancer most frequently metastasizes to the bone and skin. However, these locations are not as common in TNBC. Skin metastases signify severe systemic disease with penetration of the cancer into the skin, blood vessels, and lymphatic system [3]. According to Pogoda et al., the most common locations for recurrent TNBC were the brain and lungs, occurring in 15% and 14% of patients, respectively. In 11% of patients, metastasis occurred in the bone with a peak incidence in the second year after treatment [4]. Most bone metastases are seen in the spine and only 3.1–5.2% of patients have sternal involvement [5] [6]. When lymphangitic lung or bone marrow metastases, carcinomatous meningitis, or substantial liver metastases are present, the term visceral crisis is used [7]. The NCCN guidelines then mandate further chemotherapy based treatment. A diagnosis of TNBC can be made with morphological imaging and immunohistochemistry (IHC). Ultrasound and mammography can be used to visualize the necrosis and fibrosis that are characteristic of TNBC, and IHC detects ER, PR and HER2 status. Specimens are considered positive with 1% or more of immunoreactive tumor cells [8]. The management of TNBC is based off NCCN guidelines. Hormone receptor negative and HER2 negative disease is first divided into node positive or node negative breast cancer. Patients with node negative TNBC and tumors greater than 1 cm, like our patient, are recommended to undergo adjuvant chemotherapy. | ||||||
|
Conclusion
| ||||||
|
Our patient presented with pathologic fractures to the sternum and manubrium, nodules over the chest wall, and metastases to the lungs secondary to recurrent invasive ductal carcinoma. Triple negative breast cancer (TNBC) metastasizes most frequently to the brain and lungs, rather than the bone and skin more common of receptor positive breast cancer. Distant spread is seen more often and earlier in TNBC. The dissimilar locations of metastasis direct the need to image differently as well as early. This indicates the need for a different monitoring protocol for TNBC. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Sabrina S. Sam – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Karel T.S. Valenta – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2016 Sabrina S. Sam et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|